1
answer
0
watching
849
views
18 Feb 2019
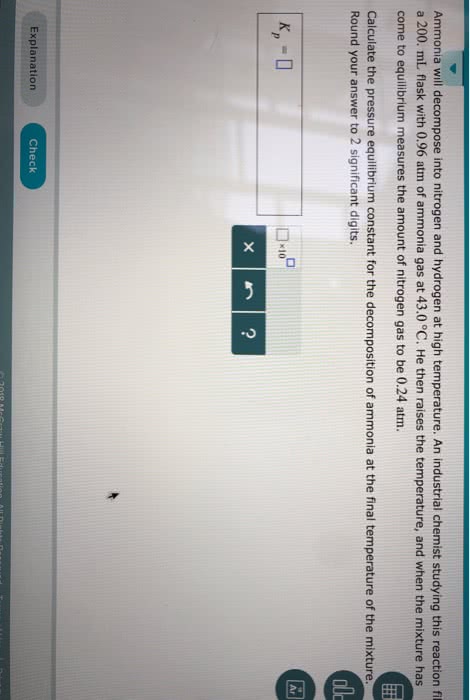
Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist studying this reaction fills a 5.0L flask with 1.7atm of ammonia gas at 51.0°C. He then raises the temperature, and when the mixture has come to equilibrium measures the amount of nitrogen gas to be 0.34atm.
.
Calculate the pressure equilibrium constant for the decomposition of ammonia at the final temperature of the mixture. Round your answer to
significant digits.
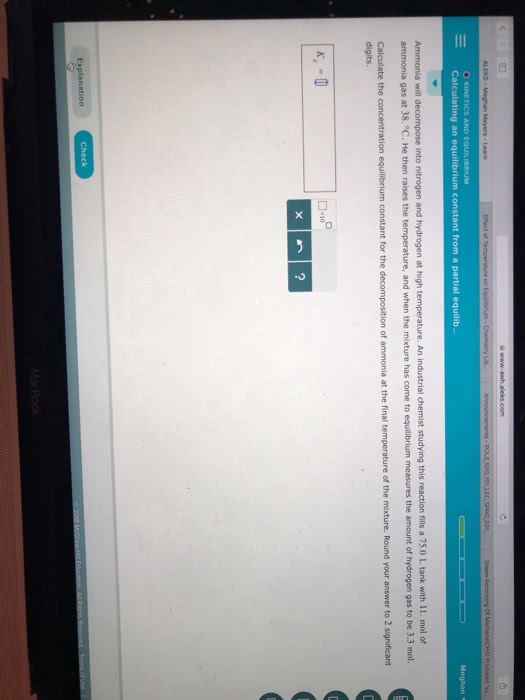
Ammonia will decompose into nitrogen and hydrogen at high temperature. An industrial chemist studying this reaction fills a 5.0L flask with 1.7atm of ammonia gas at 51.0°C. He then raises the temperature, and when the mixture has come to equilibrium measures the amount of nitrogen gas to be 0.34atm.
Calculate the pressure equilibrium constant for the decomposition of ammonia at the final temperature of the mixture. Round your answer to
Keith LeannonLv2
19 Feb 2019