1
answer
0
watching
371
views
6 Nov 2019
I performed an experiment doing free radical chlorination of1-chlorobutane + ABCN + SO2Cl2.
Resulted in four dichloro compunds. Please see question andpicture:
Four peaks representing the constitutional isomers of the reactionproducts from this experiment
will be found by GC analysis (See Picture).
QUESTION: How many total isomers (constitutional isomers plussteroisomers) are actually produced in this reaction?
List and identify them by chemical formula.
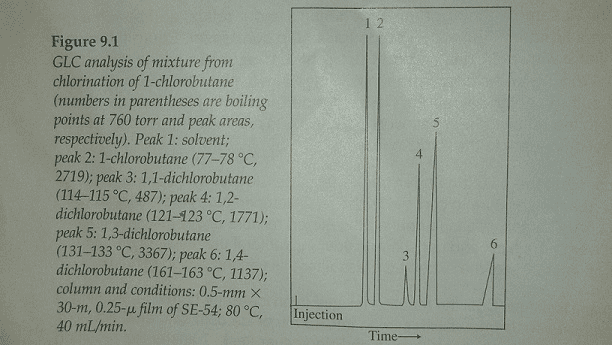
GLC analysis of mixture from chlorination of 1-chlorobutane (numbers in parentheses are boiling points at 760 torr and peak areas, respectively). Peak 1: solvent; peak 2:1-chlorobutane (77-78 degree C, 2719); peak 3; 1,1-dichlorobutane (114-115 X, 487); peak 4:1,2-dichlorobutane (121-123 degree C, 1771); peak 5:1,3-dichlorobutane (131-133 degree C, 3367); peak 6; 1,4-dichlorobutane (161-163 degree C, 1137); column and conditions: 0.5-mm times 30-m, 0.25-v film of SE-54; 80 degree C, 40 mL/min. Show transcribed image text
I performed an experiment doing free radical chlorination of1-chlorobutane + ABCN + SO2Cl2.
Resulted in four dichloro compunds. Please see question andpicture:
Four peaks representing the constitutional isomers of the reactionproducts from this experiment
will be found by GC analysis (See Picture).
QUESTION: How many total isomers (constitutional isomers plussteroisomers) are actually produced in this reaction?
List and identify them by chemical formula.

GLC analysis of mixture from chlorination of 1-chlorobutane (numbers in parentheses are boiling points at 760 torr and peak areas, respectively). Peak 1: solvent; peak 2:1-chlorobutane (77-78 degree C, 2719); peak 3; 1,1-dichlorobutane (114-115 X, 487); peak 4:1,2-dichlorobutane (121-123 degree C, 1771); peak 5:1,3-dichlorobutane (131-133 degree C, 3367); peak 6; 1,4-dichlorobutane (161-163 degree C, 1137); column and conditions: 0.5-mm times 30-m, 0.25-v film of SE-54; 80 degree C, 40 mL/min.
Show transcribed image text Irving HeathcoteLv2
15 Jul 2019
