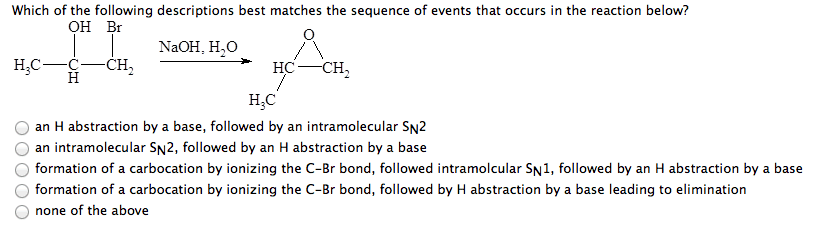
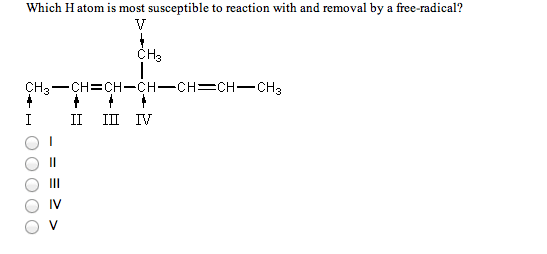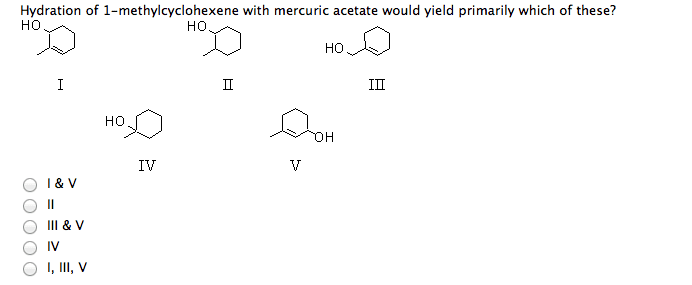Please explain how you arrived at your answers if possible &thank you in advance! :)
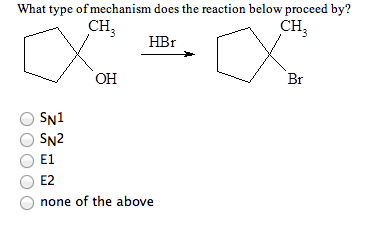
1 What mechanism does the reaction1,1-methylcyclopentanol + HBr = 1-bromo-1-methylcyclopentanolproceed by?
**I think the answer is either SN1 or SN2 but I'm not 100% sureon which one of those to choose.
a) SN1
b) SN2
c) EI
d) E2
e) none of the above
2 Which H atom is most susceptible to reaction withand removal by a free radical?
**there should be a single bond connecting the bolded carbon tothe CH3 group labeled as "V" above it
V
CH3
CH3%u2014CH =CH%u2014CH%u2014CH = CH%u2014CH3
I II III IV
a) I
b) II
c) III
d) IV
e) V
3 How many nodes,(other than the node through all p orbitals) are found inthe highest occupied molecular orbital of1,3,5-hexatriene?
a) 0
b) 1
c) 2
d) 3
e) more than 3
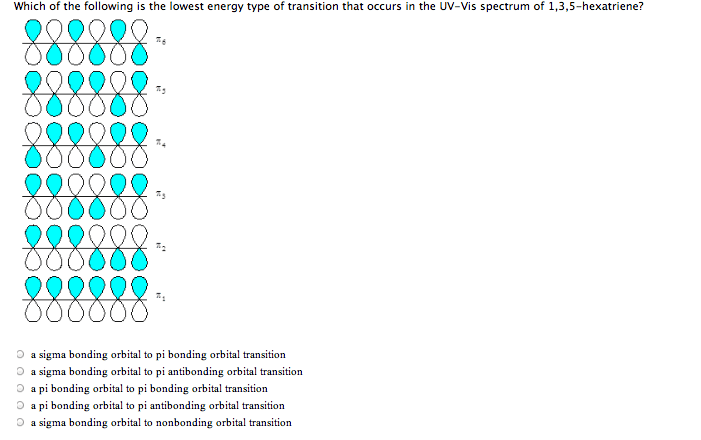
4 Which of thefollowing is the lowest energy type of transition that occurs inthe UV-Vis spectrum of 1,3,5-hexatriene?
a) a sigma bonding orbital toa pi bonding orbital transition
b) a sigma bonding orbital toa pi antibonding orbital transition
c) a pi bonding orbital to pibonding orbital transition
d) a pi bonding orbital to api antibonding orbital
e) a sigma bonding orbital toa nonbonding orbital transition
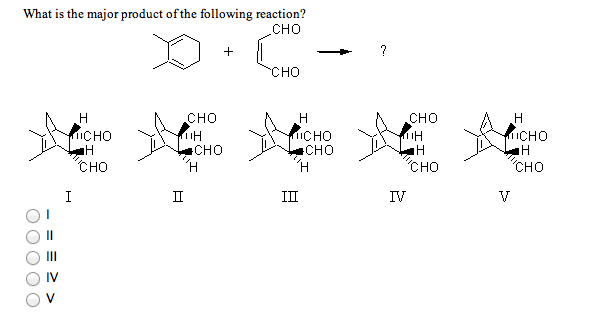
5 What is/are themajor product(s) yielded by hydration of 1-methylcyclohexene withmurcuric acetate?
Please explain how you arrived at your answers if possible &thank you in advance! :)
1 What mechanism does the reaction1,1-methylcyclopentanol + HBr = 1-bromo-1-methylcyclopentanolproceed by?
**I think the answer is either SN1 or SN2 but I'm not 100% sureon which one of those to choose.
a) SN1
b) SN2
c) EI
d) E2
e) none of the above
2 Which H atom is most susceptible to reaction withand removal by a free radical?
**there should be a single bond connecting the bolded carbon tothe CH3 group labeled as "V" above it
V
CH3
CH3%u2014CH =CH%u2014CH%u2014CH = CH%u2014CH3
I II III IV
a) I
b) II
c) III
d) IV
e) V
3 How many nodes,(other than the node through all p orbitals) are found inthe highest occupied molecular orbital of1,3,5-hexatriene?
a) 0
b) 1
c) 2
d) 3
e) more than 3
4 Which of thefollowing is the lowest energy type of transition that occurs inthe UV-Vis spectrum of 1,3,5-hexatriene?
a) a sigma bonding orbital toa pi bonding orbital transition
b) a sigma bonding orbital toa pi antibonding orbital transition
c) a pi bonding orbital to pibonding orbital transition
d) a pi bonding orbital to api antibonding orbital
e) a sigma bonding orbital toa nonbonding orbital transition
5 What is/are themajor product(s) yielded by hydration of 1-methylcyclohexene withmurcuric acetate?