1
answer
0
watching
233
views
pucezebra206Lv1
28 Sep 2019
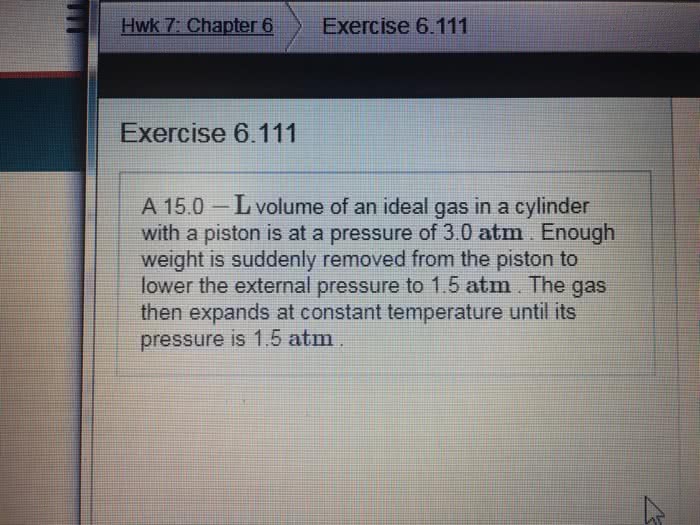
A 22.0 L volume of an ideal gas in a cylinder with a piston is at apressure of 3.0 atm. Enough weight is suddenly removed from thepiston to lower the external pressure to 1.5 atm. The gas thenexpands at constant temperature until its pressure is 1.5atm.
Find ?E for this change in state.
Find ?H for this change in state.
Find q for this change in state.
Find w for this change in state.
A 22.0 L volume of an ideal gas in a cylinder with a piston is at apressure of 3.0 atm. Enough weight is suddenly removed from thepiston to lower the external pressure to 1.5 atm. The gas thenexpands at constant temperature until its pressure is 1.5atm.
Find ?E for this change in state.
Find ?H for this change in state.
Find q for this change in state.
Find w for this change in state.
Find ?E for this change in state.
Find ?H for this change in state.
Find q for this change in state.
Find w for this change in state.
Jean KeelingLv2
28 Sep 2019