3
answers
0
watching
402
views
6 Nov 2019
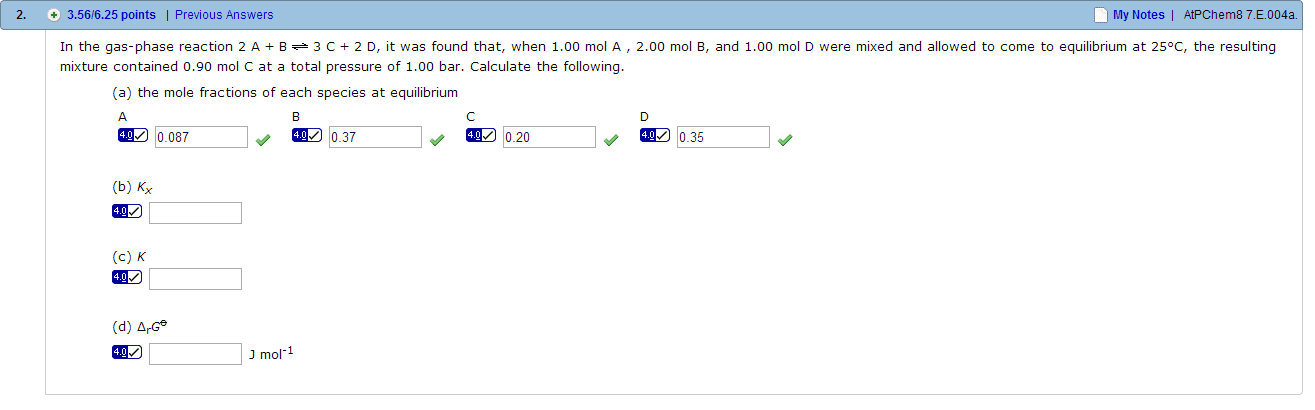
In the gas-phase reaction A + B C + 2 D, it was found that, when 2.00 mol A, 1.00 mol B, and 3.00 mol D were mixed and allowed to come to equilibrium at 25oC, the resulting mixture contained 0.79 mol C at a total pressure of 1.00 bar. Calculate (a) the mole fractions of each species at equilibrium, (b) KX, (c) K, and (d)standard Gibbs free energy
In the gas-phase reaction A + B C + 2 D, it was found that, when 2.00 mol A, 1.00 mol B, and 3.00 mol D were mixed and allowed to come to equilibrium at 25oC, the resulting mixture contained 0.79 mol C at a total pressure of 1.00 bar. Calculate (a) the mole fractions of each species at equilibrium, (b) KX, (c) K, and (d)standard Gibbs free energy
Read by 1 person
Read by 1 person
1 Mar 2023
Already have an account? Log in
Casey DurganLv2
11 Apr 2019
Already have an account? Log in