1
answer
0
watching
154
views
6 Nov 2019
Top image is Lewis structure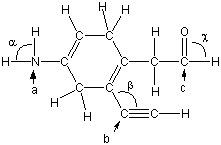
Note that lone pairs are not included in the followingstructure, but C, N, and O all obey the octet rule. Determine thelabeled bond angles and the hybridizations of the labeled atoms. Donothing to indicate a superscript. For example, write sp3 forsp3.
What are the bond angles for:
Alpha?
Beta?
Gamma?
Hybridizations for:
a?
b?
c?
Top image is Lewis structure
Note that lone pairs are not included in the followingstructure, but C, N, and O all obey the octet rule. Determine thelabeled bond angles and the hybridizations of the labeled atoms. Donothing to indicate a superscript. For example, write sp3 forsp3.
What are the bond angles for:
Alpha?
Beta?
Gamma?
Hybridizations for:
a?
b?
c?
1
answer
0
watching
154
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Patrina SchowalterLv2
24 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232