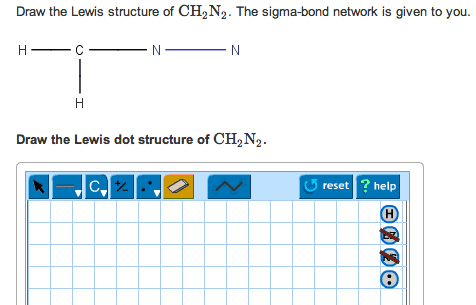
1- Draw the Lewis dotstructure of CH2N2
2-How many sp2 hybridizedcarbons are present in allene
(H2C=C=CH2)
3- The
N?H single bond in methyl amine (CH3NH2) is a ________ bond formedby the overlap of a(n) ________ orbital on Nand a(n) ________ orbital on H.
4- Which of the following is closest to the
C?O?C bond angle in CH3-O-CH3
5- Each lonepair of electrons on the
O atom in methanol (CH3OH) occupies a(n) ________orbital
6- Give the hybridization, shape, andbond angle for each carbon in
CH3CN
7- How many nonbonding electron pairs, bonding electron pairs,pi bonds, and sigma bonds are present in
CO2 ?
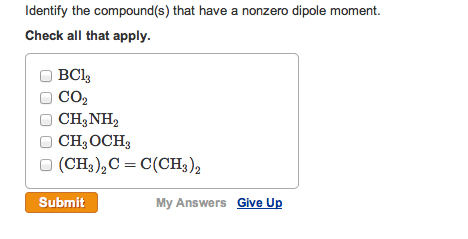
8- Identify the compound(s) that havea nonzero dipole moment.
BCl3CO2CH3NH2CH3OCH3(CH3)2C=C(CH3)2
Thanks alot.
1- Draw the Lewis dotstructure of CH2N2
2-How many sp2 hybridizedcarbons are present in allene
(H2C=C=CH2)
3- The
N?H single bond in methyl amine (CH3NH2) is a ________ bond formedby the overlap of a(n) ________ orbital on Nand a(n) ________ orbital on H.
4- Which of the following is closest to the
C?O?C bond angle in CH3-O-CH3
5- Each lonepair of electrons on the
O atom in methanol (CH3OH) occupies a(n) ________orbital
6- Give the hybridization, shape, andbond angle for each carbon in
CH3CN
7- How many nonbonding electron pairs, bonding electron pairs,pi bonds, and sigma bonds are present in
CO2 ?
8- Identify the compound(s) that havea nonzero dipole moment.
BCl3CO2CH3NH2CH3OCH3(CH3)2C=C(CH3)2
Thanks alot.
For unlimited access to Homework Help, a Homework+ subscription is required.