1
answer
0
watching
124
views
6 Nov 2019
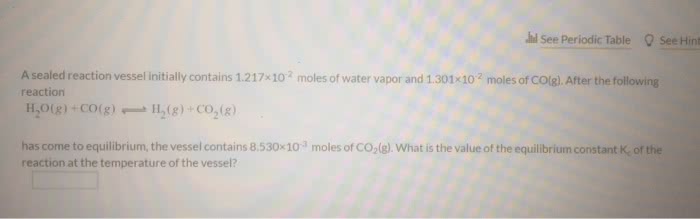
A sealed 2 L reaction vessel initially contains 1.50 x 10-2
mol of water vapor and 1.50 x
10-2
mol of CO. After the following reaction
H2O(g) + CO(g) H2(g) + CO2(g)
has come to equilibrium, the vessel contains 8.3 x 10-3
mol of CO2. What is the
equilibrium concentration of CO?
A) 3.35 x 10-3
M
B) 6.70 x 10-3
M
C) 7.50 x 10-3
M
D) 8.30 x 10-3
M
E) 1.50 x 10-2
M
A sealed 2 L reaction vessel initially contains 1.50 x 10-2
mol of water vapor and 1.50 x
10-2
mol of CO. After the following reaction
H2O(g) + CO(g) H2(g) + CO2(g)
has come to equilibrium, the vessel contains 8.3 x 10-3
mol of CO2. What is the
equilibrium concentration of CO?
A) 3.35 x 10-3
M
B) 6.70 x 10-3
M
C) 7.50 x 10-3
M
D) 8.30 x 10-3
M
E) 1.50 x 10-2
M
Keith LeannonLv2
24 Feb 2019