1
answer
0
watching
190
views
6 Nov 2019
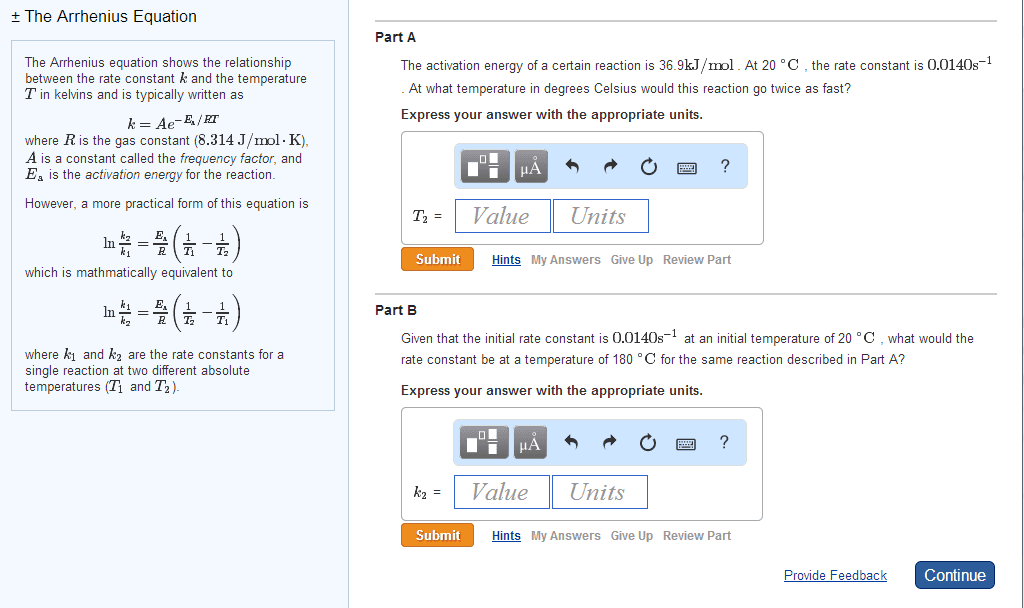
The HI decomposition reaction 2HI âãH2 + I2 Ea = 182kj/mole And at 700 degree c , the rate constant is 1.57x 10-3 M-1S-1 What is the rate constant at the 600 degree c? I use log k2/k1 = -Ea/Rx2.303 (1/T2 - 1/T1) But the answer is wrong Answer is 1.19x10-3 M-1S-1 Thanks
The HI decomposition reaction
2HI âãH2 + I2
Ea = 182kj/mole
And at 700 degree c , the rate constant is 1.57x 10-3 M-1S-1
What is the rate constant at the 600 degree c?
I use log k2/k1 = -Ea/Rx2.303 (1/T2 - 1/T1)
But the answer is wrong
Answer is 1.19x10-3 M-1S-1
Thanks
Beverley SmithLv2
22 Mar 2019