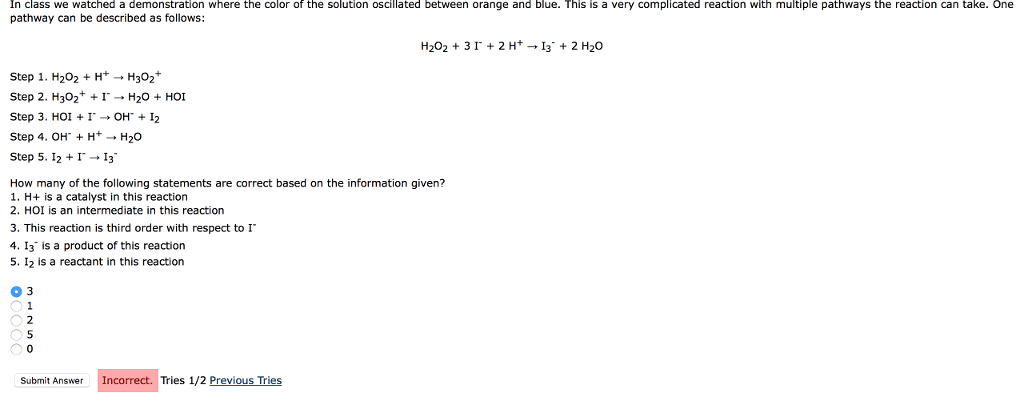In class we watched a demonstration where the color of the solution oscillated between orange and blue. This is a very complicated reaction with multiple pathways the reaction can take. One pathway can be described as follows: H2O2 + 3 I- + 2 H+ â I3- + 2 H2O
Step 1. H2O2 + H+ â H3O2+
Step 2. H3O2+ + I- â H2O + HOI
Step 3. HOI + I- â OH- + I2
Step 4. OH- + H+ â H2O
Step 5. I2 + I- â I3-
How many of the following statements are correct based on the information given?
Correct: If step 2 is the rate determining step, and step 1 is a fast equilibrium step, this reaction would be third order overall
Incorrect: There are three different intermediates in this reaction
Incorrect: An energy diagram of this reaction would have 5 peaks and the starting materials would be higher in energy than the products
Correct: There are no catalysts in this reaction
Why??
In class we watched a demonstration where the color of the solution oscillated between orange and blue. This is a very complicated reaction with multiple pathways the reaction can take. One pathway can be described as follows: H2O2 + 3 I- + 2 H+ â I3- + 2 H2O
Step 1. H2O2 + H+ â H3O2+
Step 2. H3O2+ + I- â H2O + HOI
Step 3. HOI + I- â OH- + I2
Step 4. OH- + H+ â H2O
Step 5. I2 + I- â I3-
How many of the following statements are correct based on the information given?
Correct: If step 2 is the rate determining step, and step 1 is a fast equilibrium step, this reaction would be third order overall
Incorrect: There are three different intermediates in this reaction
Incorrect: An energy diagram of this reaction would have 5 peaks and the starting materials would be higher in energy than the products
Correct: There are no catalysts in this reaction
Why??