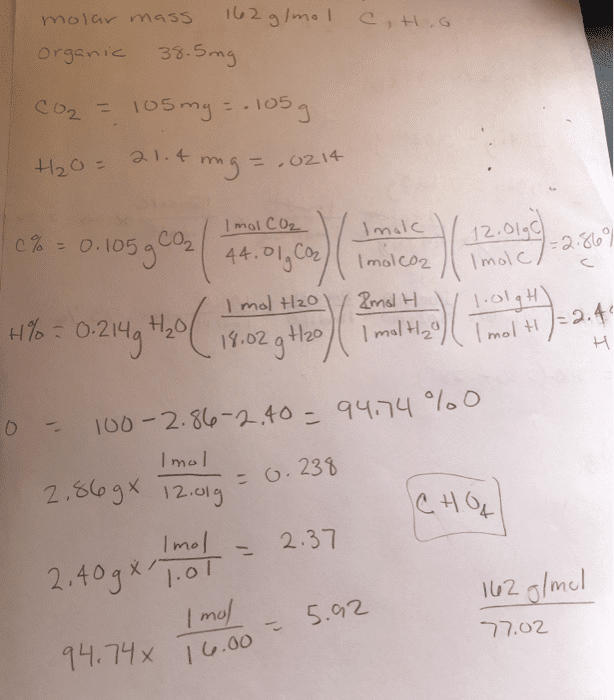1
answer
0
watching
261
views
6 Nov 2019
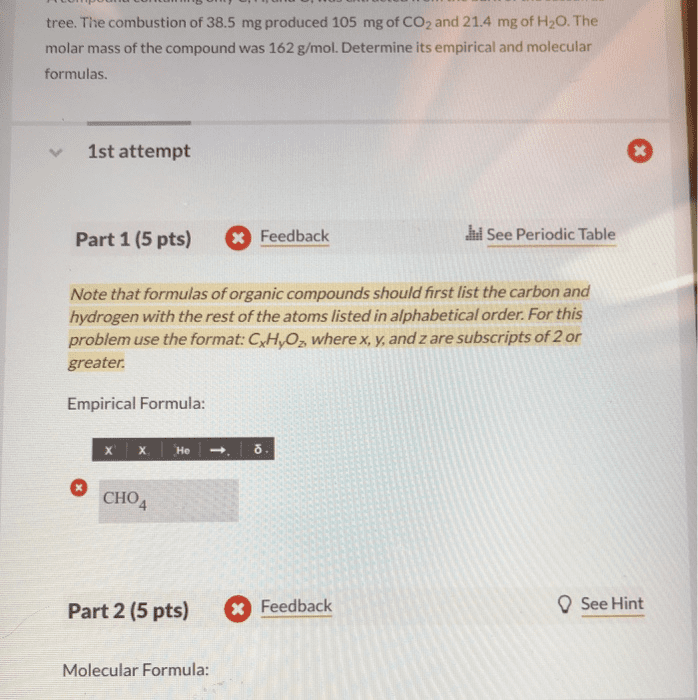
A compound containing only C, H, and O, was extracted from the bark of the sassafras tree. The combustion of 66.7 mg produced 181 mg of CO2and 37.1 mg of H2O. The molar mass of the compound was 162 g/mol. Determine its empirical and molecular formulas.
A compound containing only C, H, and O, was extracted from the bark of the sassafras tree. The combustion of 66.7 mg produced 181 mg of CO2and 37.1 mg of H2O. The molar mass of the compound was 162 g/mol. Determine its empirical and molecular formulas.
Reid WolffLv2
20 Feb 2019