1
answer
0
watching
167
views
10 Nov 2019
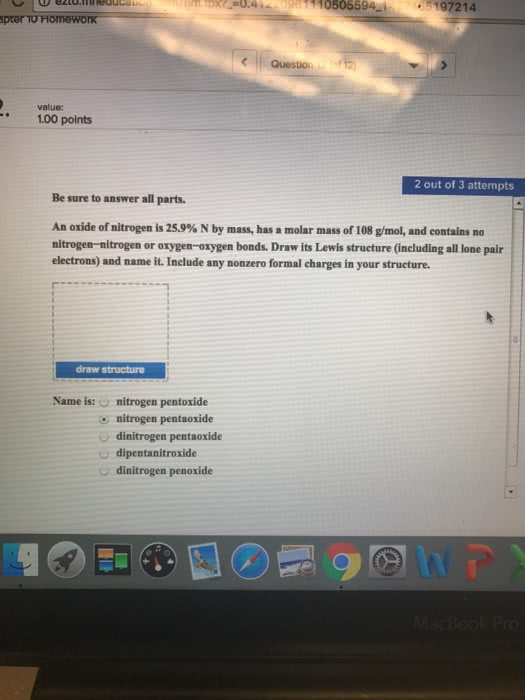
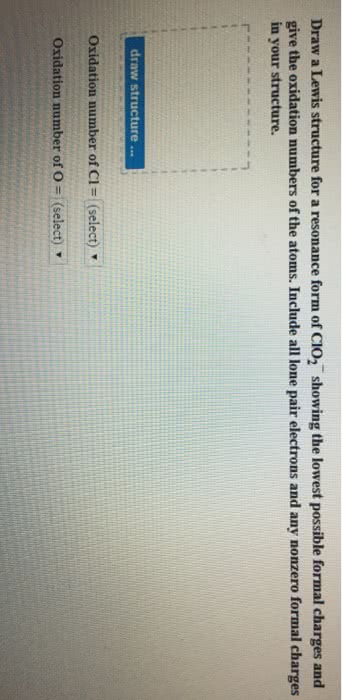
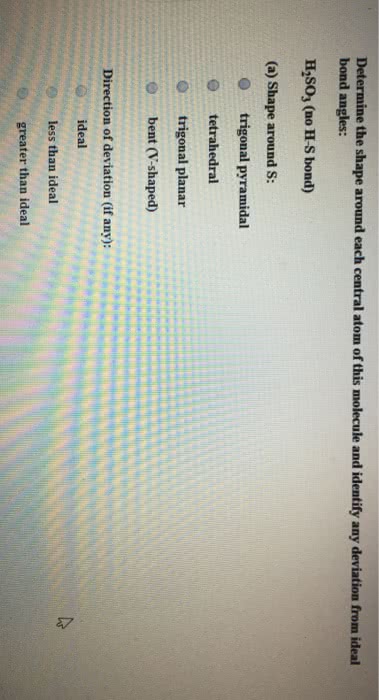
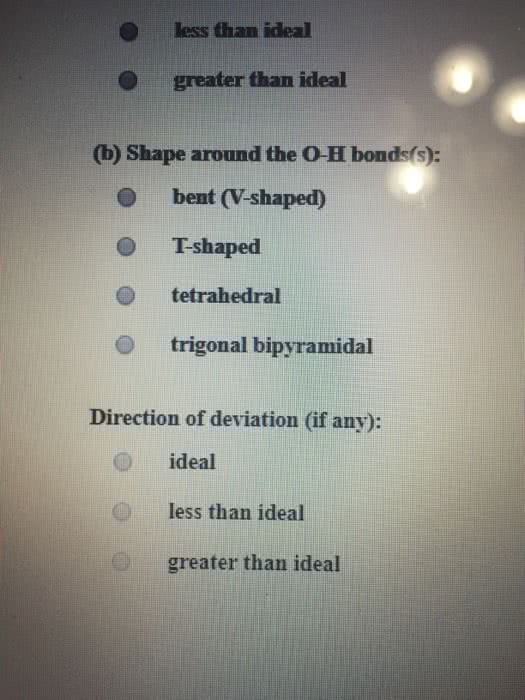
Dinitrogen monoxide (N2O) supports combustion in a manner similar to oxygen, with the nitrogen atoms forming N2. Draw one of the resonance structures for N2O (one N is central). Include all lone pair electrons and any nonzero formal charges in your structure
Dinitrogen monoxide (N2O) supports combustion in a manner similar to oxygen, with the nitrogen atoms forming N2. Draw one of the resonance structures for N2O (one N is central). Include all lone pair electrons and any nonzero formal charges in your structure
Jamar FerryLv2
30 Jan 2019