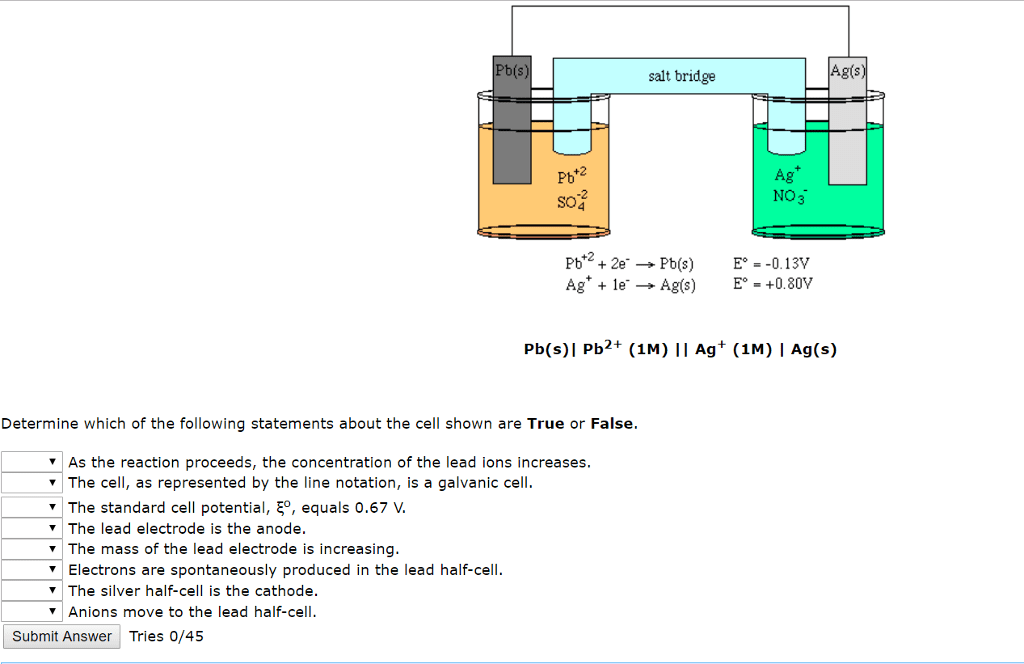
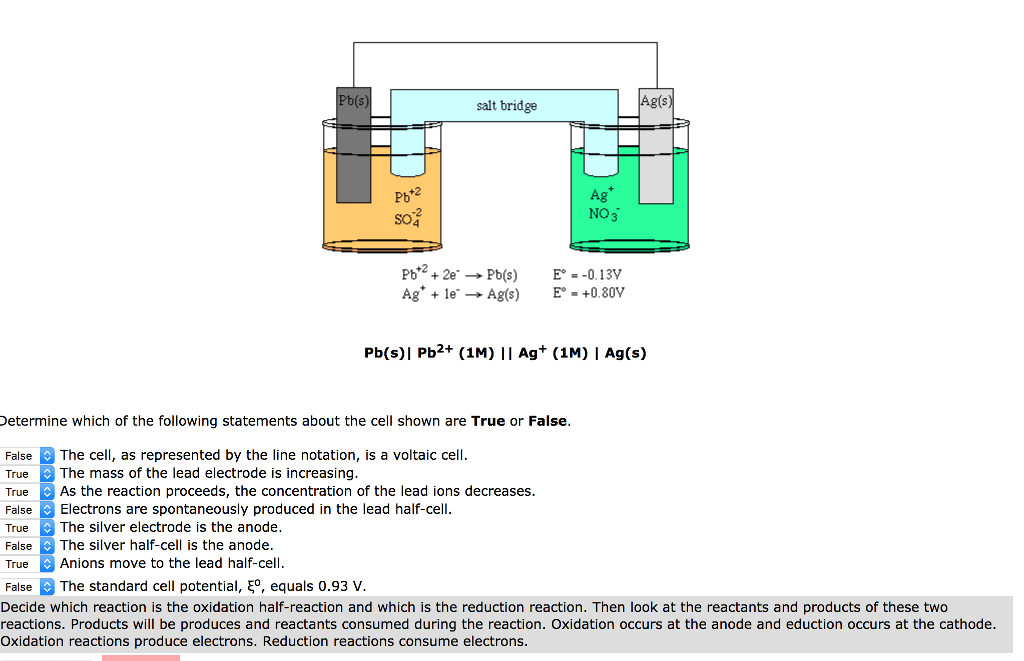
1. Write the reduction half-reaction for a Pb2+/Pb(s) half-cell.
2. Consider the construction of a voltaic cell with Pb(s)/Pb2+(aq) and Cu2+(aq)//Cu(s) half-cells.
a. If you connect the Pb(s) electrode to the red (cathode) lead and the Cu(s) electrode to the black (anode) lead and the measured Erxn shows a positive voltage, what half- cell is undergoing oxidation?
b. If you connect the Pb(s) electrode to the red (cathode) lead and the Cu(s) electrode to the black (anode) lead and the measured Erxn shows a negative voltage, what half- cell is undergoing oxidation?
3. Find the literature values for E°red of Pb2+/Pb(s) and Fe2+/Fe(s). Based on that data, predict the E°rxn for the following voltaic cell; show your work: Fe(s) | Fe2+(1M) || Pb2+(1M) | Pb(s)
4. Using the literature E°red values and applying the Nernst equation, calculate the concentration of Pb2+ needed to make the following battery: Pb(s) | Pb2+(unknown concentration) || Pb2+(1M) | Pb(s) Erxn = 0.059 V
1. Write the reduction half-reaction for a Pb2+/Pb(s) half-cell.
2. Consider the construction of a voltaic cell with Pb(s)/Pb2+(aq) and Cu2+(aq)//Cu(s) half-cells.
a. If you connect the Pb(s) electrode to the red (cathode) lead and the Cu(s) electrode to the black (anode) lead and the measured Erxn shows a positive voltage, what half- cell is undergoing oxidation?
b. If you connect the Pb(s) electrode to the red (cathode) lead and the Cu(s) electrode to the black (anode) lead and the measured Erxn shows a negative voltage, what half- cell is undergoing oxidation?
3. Find the literature values for E°red of Pb2+/Pb(s) and Fe2+/Fe(s). Based on that data, predict the E°rxn for the following voltaic cell; show your work: Fe(s) | Fe2+(1M) || Pb2+(1M) | Pb(s)
4. Using the literature E°red values and applying the Nernst equation, calculate the concentration of Pb2+ needed to make the following battery: Pb(s) | Pb2+(unknown concentration) || Pb2+(1M) | Pb(s) Erxn = 0.059 V