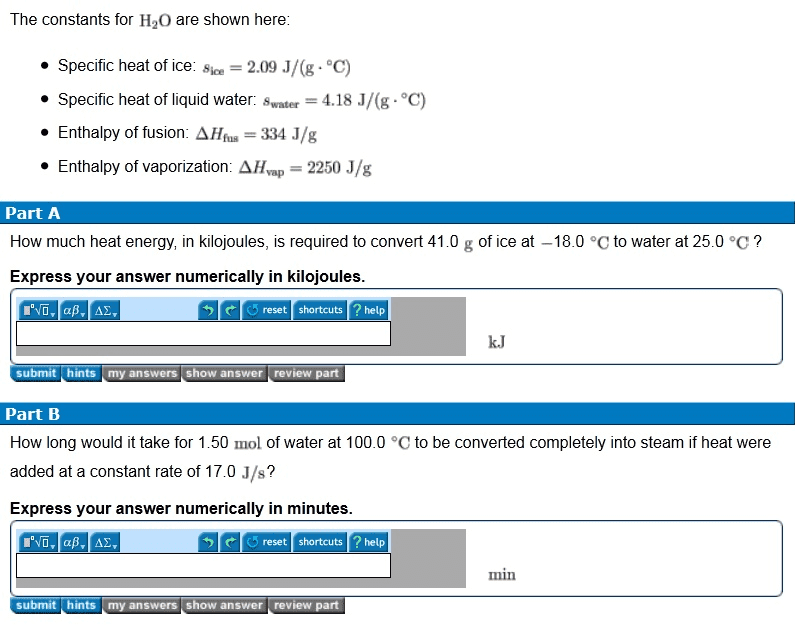
2
answers
0
watching
148
views
28 Sep 2019
A 100.0 g sample of water at 80°C, is mixed with 10 g of ice cubesat 0°C. The heat of fusion of ice is 335 J/g, and the specific heatof liquid water is 4.18 J/g°C. Calculate the final temperature indegrees Celsius of the mixture.
A. 0.0
B. 10.6
C. 30.7
D. 65
E. 53.2
A 100.0 g sample of water at 80°C, is mixed with 10 g of ice cubesat 0°C. The heat of fusion of ice is 335 J/g, and the specific heatof liquid water is 4.18 J/g°C. Calculate the final temperature indegrees Celsius of the mixture.
A. 0.0
B. 10.6
C. 30.7
D. 65
E. 53.2
A. 0.0
B. 10.6
C. 30.7
D. 65
E. 53.2
Tod ThielLv2
28 Sep 2019
Already have an account? Log in