1) A + B <--> C + DdG''(deltaGnaughtprime) = -3.1kcal/mol
2) C + D <--> E + F dG'' = 4.2kcal/mol
3) E + F <--> G + H dG'' = 0.7kcal/mol
Given this information, what would be the value ofDeltaGnaughtprime (in kcal/mol) for the followingnet reaction? A + B <--> G + H
i just don't know how to start this.
i tried labling A+B <-->C+D<--> E+F<--> G+H andtried to get rid of the C+D<-->E+F
but stuck there.
* do i multiply them all? and get (-)9.114?
its got to be one of these +1.8, -1.8, -9.1 ,9.1
1) A + B <--> C + DdG''(deltaGnaughtprime) = -3.1kcal/mol
2) C + D <--> E + F dG'' = 4.2kcal/mol
3) E + F <--> G + H dG'' = 0.7kcal/mol
Given this information, what would be the value ofDeltaGnaughtprime (in kcal/mol) for the followingnet reaction? A + B <--> G + H
i just don't know how to start this.
i tried labling A+B <-->C+D<--> E+F<--> G+H andtried to get rid of the C+D<-->E+F
but stuck there.
* do i multiply them all? and get (-)9.114?
its got to be one of these +1.8, -1.8, -9.1 ,9.1
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
1-How many grams of NaCl would be needed to prepare 150.00 L of a 1.402 M NaCl solution?
............................................................................................................................................................................
2- first a standardized solution of sodium hydroxide (NaOH) will be quantitatively diluted to about 0.1 M but not exactly 0.1M. The molarity of the dilute NaOH will be calculated! A similar quantitative dilution was done in experiment 3 when you diluted 5.0 M HCl to 0.50M HCl using a volumetric flask and pipet.
The second part of the experiment is very similar to the last part of experiment 6 where samples of the unknown were weighed out, dissolved in water, and titrated with the dilute NaOH using phenolphthalein as the indicator to determine the percent by mass of a compound in the unknown. The compound will be different than the compound used in experiment 6. The molecular formula of the compound will be provided during the final. You must know how to weigh by difference (Preweigh, tare sample and small container to zero, transfer sample to flask, and place the small container back on balance to get mass) and titrate the sample using the buret in order to do well.
Be sure to read the instructions for the practical toward the end the CHM151L lab manual and do the practice problem on the next page.
You will have 1.5 hours to do the practical. You may only use a calculator and a pencil on the practical. There is no talking during the practical such that students cannot help each other and the TA may only help you with equipment problems.
It would be a good idea to wash any glassware you might use the lab period before the lab practical. There will also be a help session before the lab practical.
At the end of the lab final you will wash all glassware used or any dirty glassware in your locker, wash your unknown vials and remove the labels, checkin your locker and return your key, and evaluate the lab.
Please put the following statements in the correct order relating to the day of the lab practical:
1 2 3 4 5 6 Wash all your glassware and unknown vials, checkin your locker, evaluate the lab, and check your grades.
1 2 3 4 5 6 Do at least 3 good trials (weigh and titrate sample) as time allows.
1 2 3 4 5 6 Start the lab practical by diluting the NaOH solution.
1 2 3 4 5 6 Before the day of the practical clean glassware, read the practical instructions and do the practice problem in the lab manual and complete the loncapa problems for the final.
1 2 3 4 5 6 Calculate the molarity of the dilute NaOH, the percent by mass of the compound in you unknown for each trial and then the median.
1 2 3 4 5 6 Come to the help session before the lab final.
............................................................................................................................................................................
3-
A NaOH solution was prepared by diluting 5.00 mL of 2.205 M NaOH to a final total volume of 100.00 mL. What is the exact molarity of the dilute NaOH (use 4-5 significant figures)?
............................................................................................................................................................................
4-For the problem above, select glassware and size from the list below to conduct the dilution.
| A | Amber Bottle |
| B | Mohr pipet |
| C | Buret |
| D | Volumetric Flask |
| E | Volumetric Pipet or Pipettor |
| F | Graduated cylinder |
| G | Erlenmeyer flask |
| H | Beaker |
A B C D E F G H <= Glassware used to transfer the 2.205 M NaOH to be diluted
A B C D E F G H <= Glassware used to measure the final volume of the diluted M NaOH.
5 10 15 20 25 50 100 250 500 1000 <= Volume of the glassware chosen to transfer the concentrated NaOH above
5 10 15 20 25 50 100 250 500 1000 <= Volume of the glassware chosen to contain the final dilute NaOH above.
Everytime I make the graph based off my calculations in the chart it looks like this and I have no idea if it is even close? Please help ! For the questions I just need to understand how to start them and what info is nessiary to do so. very confused. 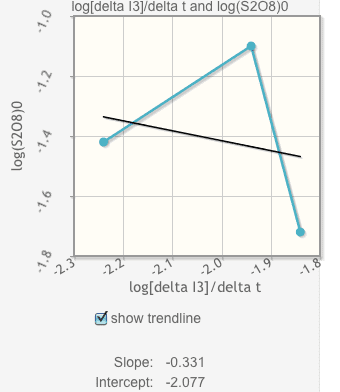
Data Analysis
Fill in the following table. Look below the table for instructions on how to calculate the values for each row of the table.
| a | total volume (L) | 0.065L |
| b | mol S2O32-consumed | 0.010 mol |
| c | mol I3-produced | 0.015M |
| d | [S2O32-] consumed (M) | 0.154M |
| e | [I3-] produced (M) | 0.231M |
Calculate the final volume of the reaction mixture after the contents of beaker B are added to beaker A. Report your answer in liters.
All S2O32- is consumed at the end of the reaction. Therefore, the moles of S2O32- consumed can be calculated using the equation below.
(moles S2O32-)consumed = Mstock x (Vstock)
The I3- produced in reaction 1 reacts with S2O32- in reaction 2 as shown.
I3- (aq) 2S2O32- (aq) â 3I- (aq) S4O62- (aq)
Therefore, for every mole of I3- produced, 2 moles of S2O32- have reacted.
(moles I3-)produced = (moles S2O32-)consumed / 2
[S2O32-]consumed = (moles S2O32-)consumed / (Vtotal)
Calculation is the same as d, but using I3- instead of S2O32-.
Fill in the following table. Look below the table for instructions on how to calculate the values for each column of the table.
| a | b | c | d | e | f | g | h | i | |
|---|---|---|---|---|---|---|---|---|---|
| Trial | Ît | Î[I3-] / Ît (M/s) | log(Î[I3-] / Ît) | V 0.2 M KI added (mL) | [Iâ]0(M) | log[Iâ]0 | V of 0.2 M (NH4)S2O8 (mL) | [S2O8]0 (M) | log[S2O8]0 |
| 1 | 19 | .011 | -1.94 | 25 | .077 | -1.35 | 25 | .077 | -1.11 |
| 2 | 40 | .0055 | -2.24 | 25 | .077 | -1.11 | 12.5 | .038 | -1.42 |
| 3 | 85 | .0030 | -1.84 | 25 | .077 | -.1.11 | 6.25 | .019 | -1.72 |
| 4 | 42 | .0055 | -2.26 | 12.5 | .038 | -1.42 | 25 | .077 | -1.11 |
Conclusions
If a large amount of heat was released at the start of the reaction, what effect would this have on the rate measurements?
| The rate of reaction would be much slower due to its exothermic reaction properties. |
Click the box underneath the graph to show the trendline. It will automatically calculate your slope and intercept. Record them below.
| Determine the value of q from the graph. Explain your answer. |
What is the rate law for the reaction?
For each trial, calculate the rate constant. What is average value of the rate constant?
In this experiment, you assumed that [S2O82â] >> [S2O32â]. To find out if this assumption is correct, calculate the ratio of [S2O82â] / [S2O32â] for Trial 3. (In Trial 3, the [S2O82â] was lowest, and therefore the ratio [S2O82â] / [S2O32â] is the smallest).
In this experiment, you assumed that only a small amount of S2O82â was used during the time trial so that the concentration of this reactant did not change appreciably during the course of the reaction. Calculate how much S2O82â was used in Trial 3 and the percent remaining at the end of the reaction. Was this assumption valid?

