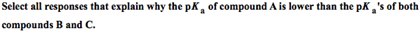1
answer
0
watching
126
views
10 Nov 2019
explain in detail why methanol is less acidic than phenol
pKa of ethanol is 15.5 and pKa of phenol is 9.95
**please try and use the actual terms like inductive effect,electronegativity, electron withdrawing group, electron donatinggroup and if there are any drawings (like of thedestabiization/stabilization of a charge) it would be greatlyappreciated!!!
thanks in advance for any help
explain in detail why methanol is less acidic than phenol
pKa of ethanol is 15.5 and pKa of phenol is 9.95
**please try and use the actual terms like inductive effect,electronegativity, electron withdrawing group, electron donatinggroup and if there are any drawings (like of thedestabiization/stabilization of a charge) it would be greatlyappreciated!!!
thanks in advance for any help
Jamar FerryLv2
22 Aug 2019