Chemistry Homework help with explanations!!
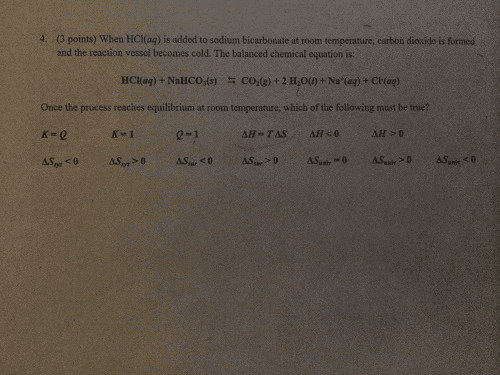
When HCl(aq) is added to sodium bicarbonate at room temperature, carbon dioxide is formed and the reaction vessel becomes cold. The balanced chemical equation is: HCl(aq) + NaHCO3(s) leftarrow rightarrow CO2(g) + 2 H2O(l) + Na+(aq) + Cl(aq) Once the process reaches equilibrium at room temperature, which of the following must be true?
Chemistry Homework help with explanations!!
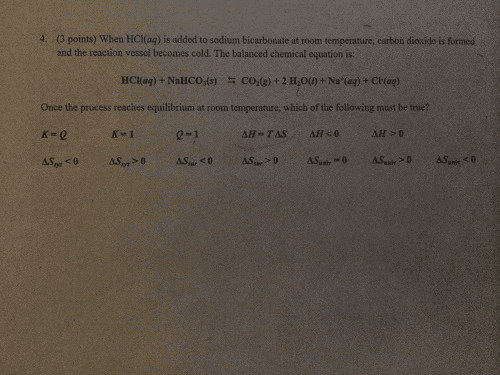
For unlimited access to Homework Help, a Homework+ subscription is required.
Related textbook solutions
Basic Chemistry
Principles of Chemistry Molecular Approach
Chemistry: Structure and Properties
Principles of Chemistry Molecular Approach
Chemistry: A Molecular Approach
Chemistry: A Molecular Approach
Principles of Chemistry: A Molecular Approach
Chemistry: The Central Science
Related questions
Assignment 5: Separation Scheme Correction
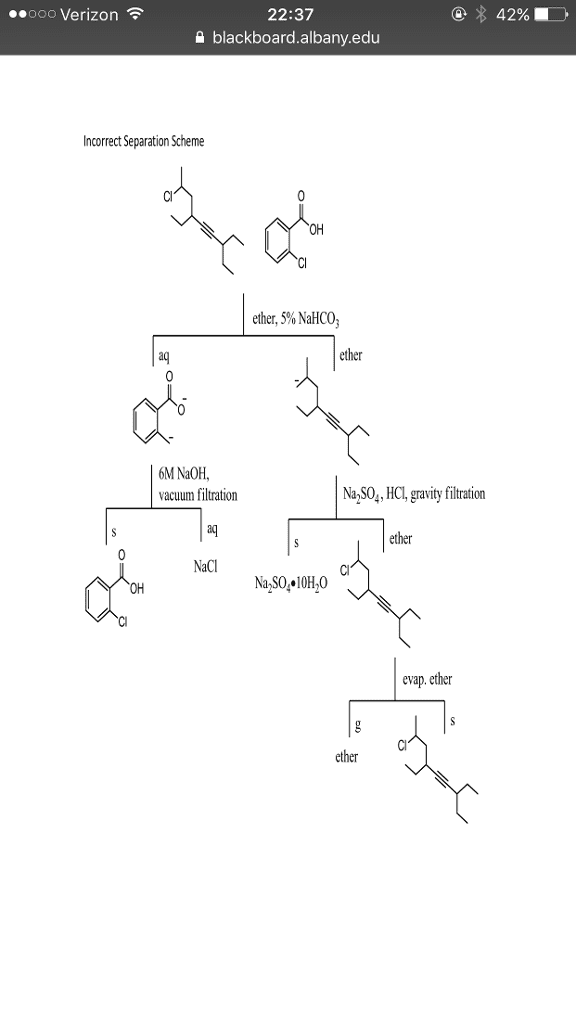
ï On a page titled Incorrect Separation Scheme print the incorrect separation scheme provided for your molecule on Blackboard. The top of the separation scheme shows what other compound is mixed with your molecule. Assume for the purposes of this assignment that both compounds are solid at room temperature. Also assume that both compounds are soluble in ether, except ionic compounds. The goal of the separation is the isolate each of the two compounds from the mixture.
o Below the incorrect separation scheme write a discussion of this incorrect scheme identifying all of the mistakes in the separation scheme. Keep in mind that there will be more than one mistake in the scheme.
o For each mistake, give a detailed, scientific explanation of why it is incorrect. o All answers must be typed. No handwritten responses will be accepted.
ï On a page titled Correct Separation Scheme draw using a chemical drawing program or other computer generated form, a correct separation scheme to isolate your molecule from the provided second molecule. Make sure to indicate the phase of each chemical in every step (s, l, g, aq) and write the separation technique used in each step (ie: vacuum filtration). The list below gives the reagents available in the lab. Use any of the reagents below to isolate the compounds. Remember that the simplest separation scheme is the best one. Try to avoid unnecessary steps. No hand drawn structures will be accepted.
| ï· 10% aqueous NaOH | ï· H2O |
| ï· 5% aqueous NaHCO3 | ï· H3PO4 |
| ï· 6M HCl | ï· anhydrous Na2SO4 |
| ï· 5% HCl | ï· anhydrous MgSO4 |
| ï· Diethyl ether (ether) | ï· Saturated NaCl |
| ï· CH3OH | ï· Sodium borohydride (NaBH4) |
| ï· lithium aluminum hydride (LiAlH4) | ï· NaCl solid |
