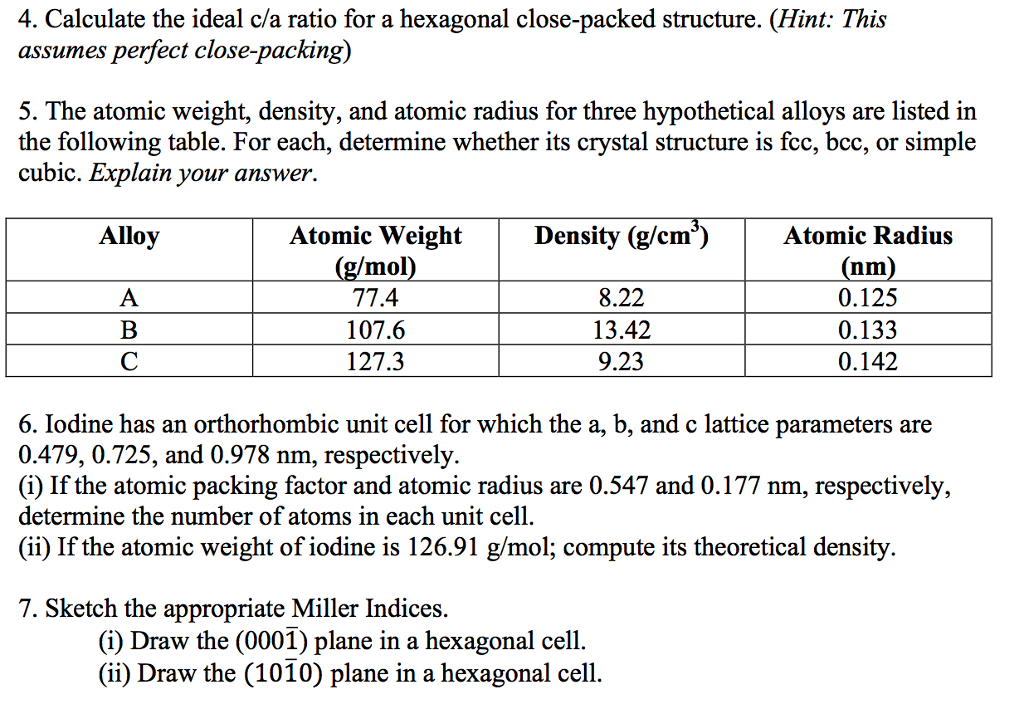
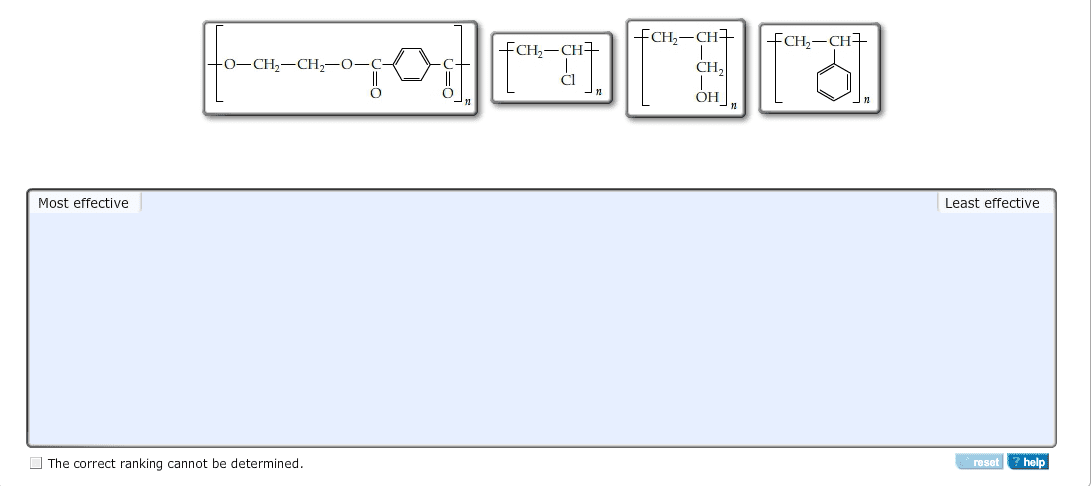
1
answer
0
watching
1,833
views
10 Nov 2019
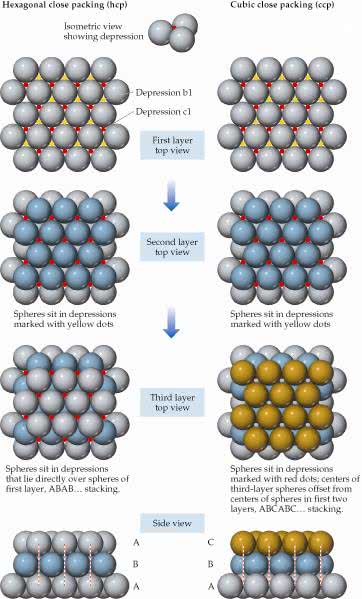

Close packing ofequal-sized spheres. Hexagonal (left) close packing and cubic(right) close packing are equally efficient ways of packingspheres. The unit cells for (a) ahexagonal close-packed metal and (b) a cubic close-packed metal.The solid lines indicate the unit cell boundaries.
What type of two-dimensional lattice describes thestructure of a single layer of close-packed atoms?
 |  |
| Close packing ofequal-sized spheres. Hexagonal (left) close packing and cubic(right) close packing are equally efficient ways of packingspheres. | The unit cells for (a) ahexagonal close-packed metal and (b) a cubic close-packed metal.The solid lines indicate the unit cell boundaries. What type of two-dimensional lattice describes thestructure of a single layer of close-packed atoms? |
Jarrod RobelLv2
17 Jun 2019