1
answer
0
watching
285
views
10 Nov 2019
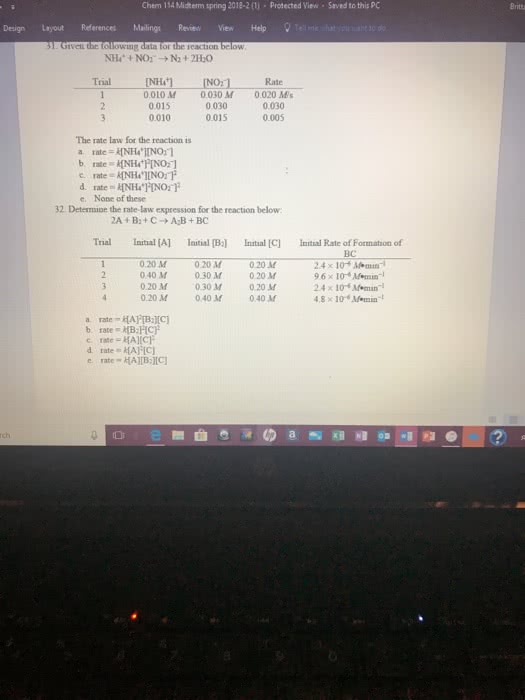
Given the following experimental data, find the rate law and therate constant for the reaction:
NO(g)+NO2(g)+O2(g) *arrow* N2O5(g)
Run [NO], M [NO2], M [O2], M Initial Rate, Ms-1
1 0.10 M 0.10M 0.10M 2.1*10^-2
2 0.20 M 0.10M 0.10M 4.2*10^-2
3 0. 20 M 0.30 M 0.20M 1.26*10^-1
4 0.10 M 0.10 M 0.20M 2.1*10^-2
please show all steps
Given the following experimental data, find the rate law and therate constant for the reaction:
NO(g)+NO2(g)+O2(g) *arrow* N2O5(g)
Run [NO], M [NO2], M [O2], M Initial Rate, Ms-1
1 0.10 M 0.10M 0.10M 2.1*10^-2
2 0.20 M 0.10M 0.10M 4.2*10^-2
3 0. 20 M 0.30 M 0.20M 1.26*10^-1
4 0.10 M 0.10 M 0.20M 2.1*10^-2
please show all steps
Patrina SchowalterLv2
19 May 2019