1
answer
2
watching
1,433
views
10 Nov 2019
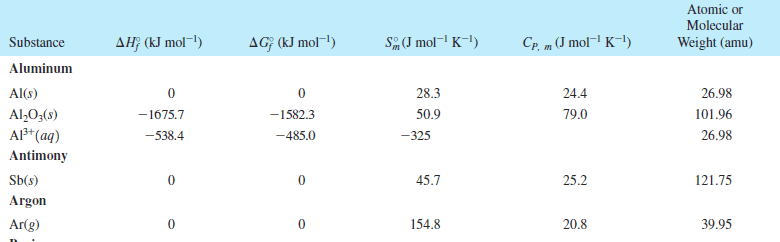
7. (20 pts.) A sample of 1.00 mol of perfect gas molecules, originally at 1.00 atm, with a molar heat capacity at constant pressureof 7/2 R is put through the following cycle:a. constant volume heating to twice its initial temperature, where ?T = 298 K,b. reversible, adiabatic expansion back to its initial temperature,c. reversible, isothermal compression back to 1.00 atm. Calculate q, w, ?U, and ?H for each step and for the overall processPlease show work/equations Thanks
7. (20 pts.) A sample of 1.00 mol of perfect gas molecules, originally at 1.00 atm, with a molar heat capacity at constant pressureof 7/2 R is put through the following cycle:a. constant volume heating to twice its initial temperature, where ?T = 298 K,b. reversible, adiabatic expansion back to its initial temperature,c. reversible, isothermal compression back to 1.00 atm. Calculate q, w, ?U, and ?H for each step and for the overall processPlease show work/equations Thanks
Liked by malaknaji47
Jarrod RobelLv2
9 May 2019