1
answer
0
watching
164
views
10 Nov 2019

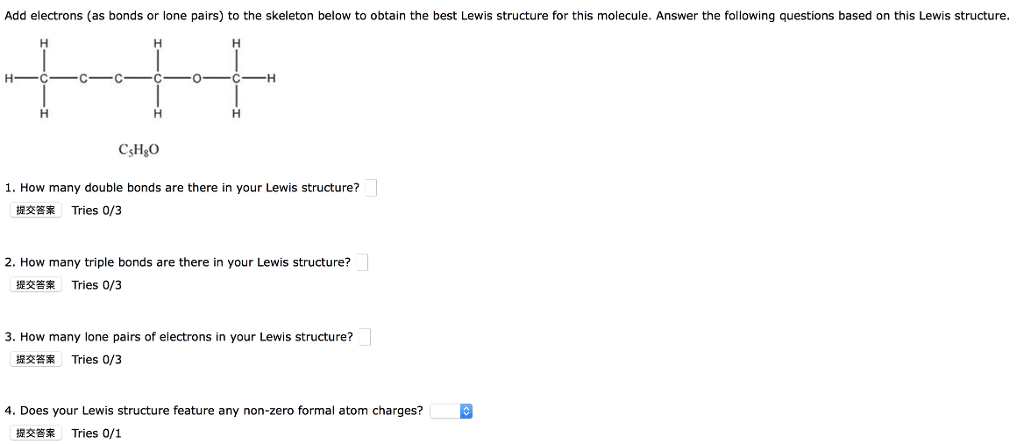
Finish drawing the Lewis structures of the following molecules and then tell how many sigma and pi bonds and how many bond pairs and lone pairs there are in each. 2. Based on formal charge assignment of the atoms, which of the following is the best Lewis structure for the bromate ion, BrO3?Chem|3|4- Year 2014/2015

Finish drawing the Lewis structures of the following molecules and then tell how many sigma and pi bonds and how many bond pairs and lone pairs there are in each. 2. Based on formal charge assignment of the atoms, which of the following is the best Lewis structure for the bromate ion, BrO3?Chem|3|4- Year 2014/2015
1
answer
0
watching
164
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Hubert KochLv2
4 Oct 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232