1
answer
0
watching
122
views
10 Nov 2019
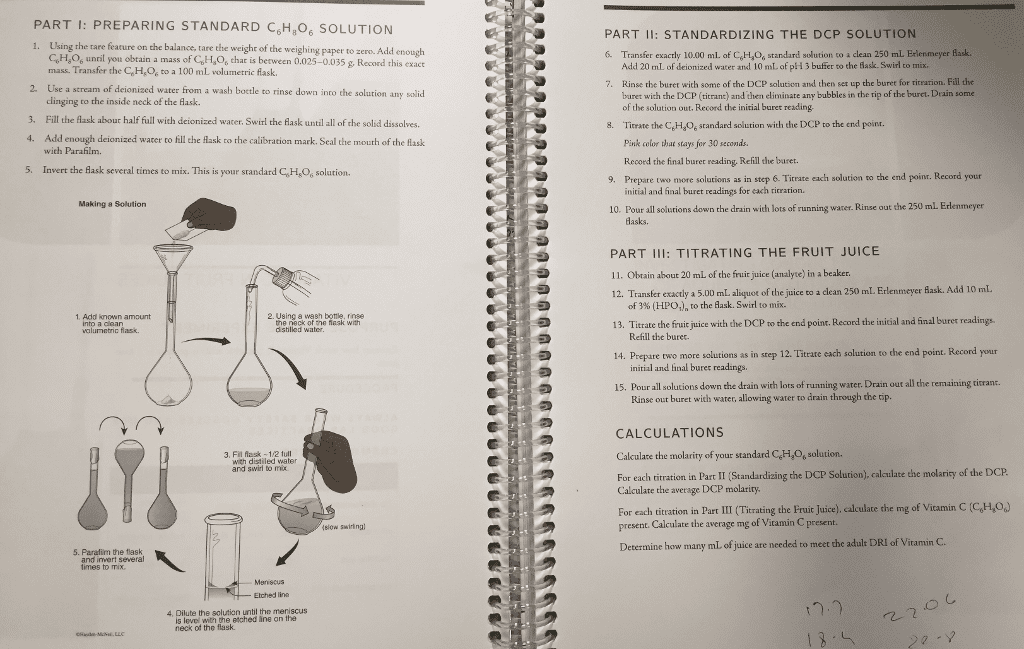
1.700 grams of baking soda are rinsed into a 100.00 mLvolumetric flask with distilled water. Then the flask is filled tothe mark with distilled water and shaken until all solid isdissolved. A 10.00 mL sample of the resulting solution istransferred into an Erlenmeyer flask for a titration. Upontitration you determine that there are 0.0340 grams of NaHCO3present in the sample. What is the percent purity of NaHCO3 in yourbaking soda? (The formula weight of NaHCO3 is 84.007 g/mole).
1.700 grams of baking soda are rinsed into a 100.00 mLvolumetric flask with distilled water. Then the flask is filled tothe mark with distilled water and shaken until all solid isdissolved. A 10.00 mL sample of the resulting solution istransferred into an Erlenmeyer flask for a titration. Upontitration you determine that there are 0.0340 grams of NaHCO3present in the sample. What is the percent purity of NaHCO3 in yourbaking soda? (The formula weight of NaHCO3 is 84.007 g/mole).
1
answer
0
watching
122
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Beverley SmithLv2
17 Jul 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232