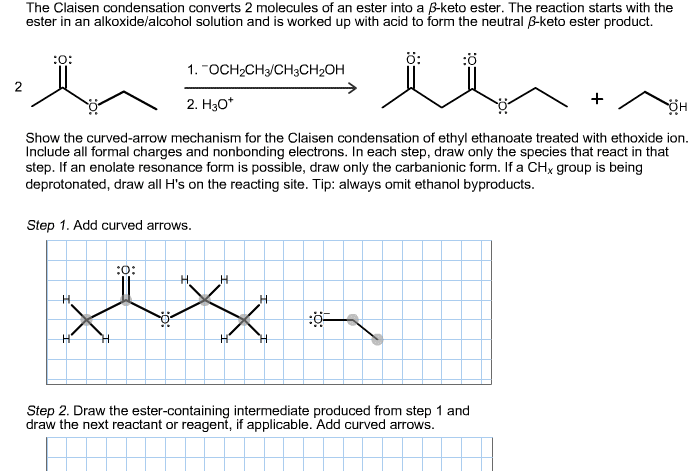
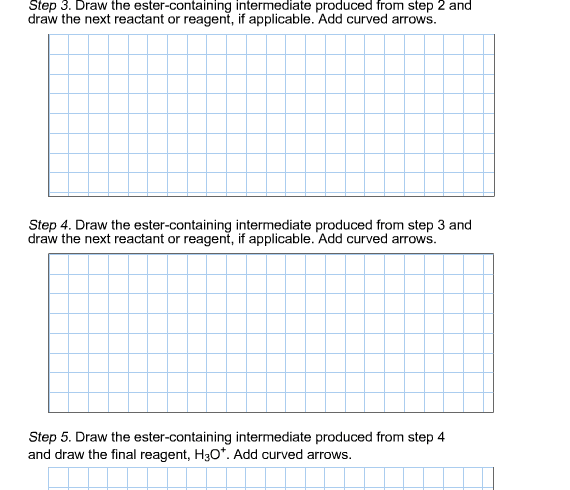
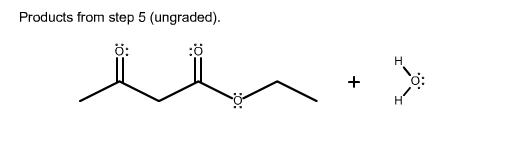
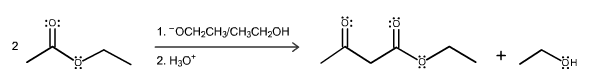1
answer
0
watching
338
views
11 Nov 2019
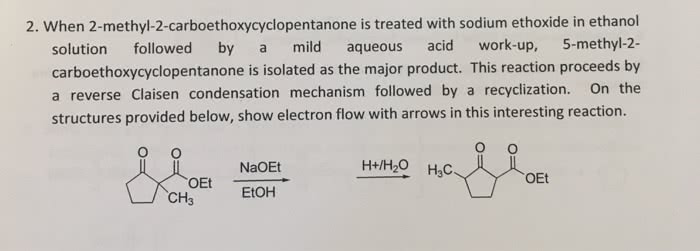

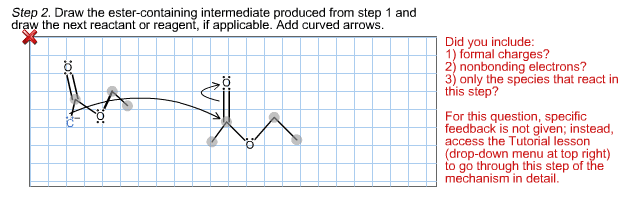
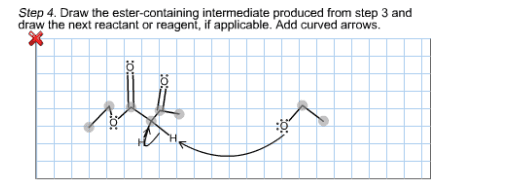
When 2-methyl-2-carboethoxycyclopentanone is treated with sodium ethoxide in ethanol solution followed by a mild aqueous acid work-up, 5-methyl-2-carboethoxycyclopentanone is isolated as the major product. This reaction proceeds by a reverse Claisen condensation mechanism followed by a recyclization. On the structures provided below, show electron flow with arrows in this interesting reaction.
1
answer
0
watching
338
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Irving HeathcoteLv2
7 Jul 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232