1
answer
0
watching
92
views
11 Nov 2019
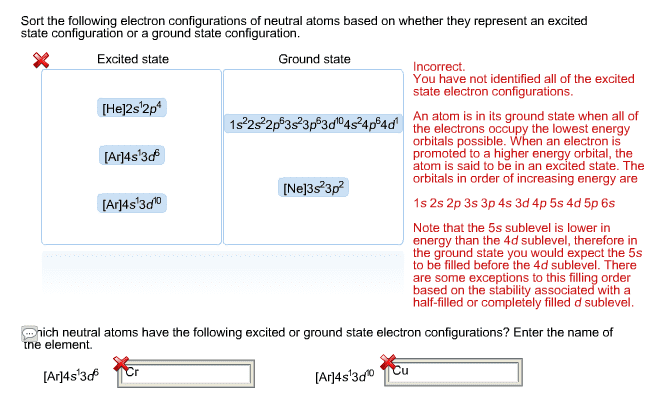
In nature, there are many exceptions to the basic order-of-filling rules to describe electron configuration. For example, the electron configuration of copper is found experimentally to be [Ar]3d104s1, rather than the configuration of [Ar]4s23d9 predicted by the standard rules. Why would the experimentally determined configuration to differ from that predicted by the standard rules? (Note that the electron configuration of Ni is: [Ar]4s23d8.) Based on the experimentally determined configuration, what shape would you expect a copper atom to have?
In nature, there are many exceptions to the basic order-of-filling rules to describe electron configuration. For example, the electron configuration of copper is found experimentally to be [Ar]3d104s1, rather than the configuration of [Ar]4s23d9 predicted by the standard rules. Why would the experimentally determined configuration to differ from that predicted by the standard rules? (Note that the electron configuration of Ni is: [Ar]4s23d8.) Based on the experimentally determined configuration, what shape would you expect a copper atom to have?
1
answer
0
watching
92
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Deanna HettingerLv2
10 May 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232