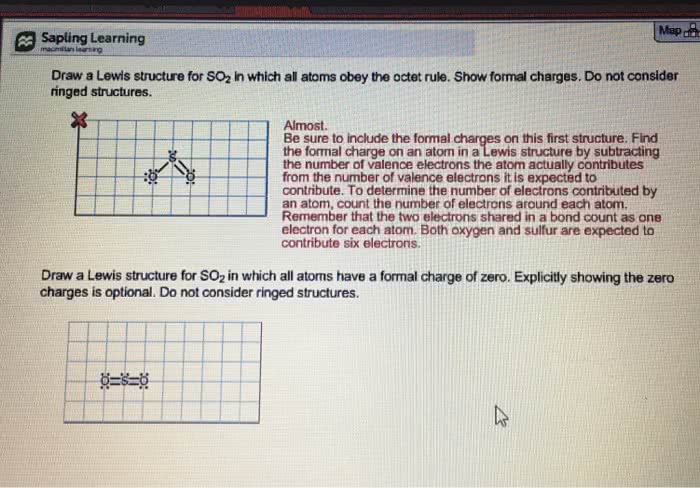
1
answer
0
watching
884
views
11 Nov 2019
a) Draw Lewis structures for NS2+ where N is the central atom. Restrict yourself to structures in which the formal charges are consistent with the relative electronegativities of the elements. Assuming idealized angles, predict the S-N-S bond angle.
b) The Lewis structure for NS2+ that corresponds to the experimental structure has a +1 formal charge on N. Assuming idealized angles, predict the S-N-S bond angle.
a) Draw Lewis structures for NS2+ where N is the central atom. Restrict yourself to structures in which the formal charges are consistent with the relative electronegativities of the elements. Assuming idealized angles, predict the S-N-S bond angle.
b) The Lewis structure for NS2+ that corresponds to the experimental structure has a +1 formal charge on N. Assuming idealized angles, predict the S-N-S bond angle.
Trinidad TremblayLv2
3 Sep 2019