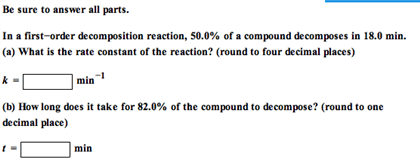1
answer
0
watching
480
views
11 Nov 2019
In a first-order decomposition reaction, 50.0% of a compound decomposes in 19.5 min.
(a) What is the rate constant of the reaction?
(b) How long does it take for 75% of the compound to decompose?
In a first-order decomposition reaction, 50.0% of a compound decomposes in 19.5 min.
(a) What is the rate constant of the reaction?
(b) How long does it take for 75% of the compound to decompose?
Jarrod RobelLv2
24 Jan 2019