1
answer
0
watching
632
views
11 Nov 2019
Solubility-Product Constants at 298 K
Compound Formula Ksp Aluminium hydroxide Al(OH)3 4.6Ã10â33 Aluminium phosphate AlPO4 9.84Ã10â21 Antimony sulfide Sb2S3 1.6Ã10â93 Arsenic sulfide As2S3 1Ã10â16 Barium bromate Ba(BrO3)2 2.43Ã10â4 Barium carbonate BaCO3 2.58Ã10â9 Barium chromate BaCrO4 1.17Ã10â10 Barium fluoride BaF2 1.84Ã10â7 Barium hydroxide octahydrate Ba(OH)2·8H2O 2.55Ã10â4 Barium iodate Ba(IO3)2 4.01Ã10â9 Barium iodate monohydrate Ba(IO3)2·H2O 1.67Ã10â9 Barium molybdate BaMoO4 3.54Ã10â8 Barium nitrate Ba(NO3)2 4.64Ã10â3 Barium phosphate Ba3(PO4)2 3.40Ã10â23 Barium selenate BaSeO4 3.40Ã10â8 Barium sulfate BaSO4 1.08Ã10â10 Barium sulfite BaSO3 5.0Ã10â10 Beryllium hydroxide Be(OH)2 6.92Ã10â22 Bismuth arsenate BiAsO4 4.43Ã10â10 Bismuth iodide BiI 7.71Ã10â19 Cadmium arsenate Cd3(AsO4)2 2.2Ã10â33 Cadmium carbonate CdCO3 1.0Ã10â12 Cadmium fluoride CdF2 6.44Ã10â3 Cadmium hydroxide Cd(OH)2 7.2Ã10â15 Cadmium iodate Cd(IO3)2 2.5Ã10â8 Cadmium oxalate trihydrate CdC2O4·3H2O 1.42Ã10â8 Cadmium phosphate Cd3(PO4)2 2.53Ã10â33 Cadmium sulfide CdS 1Ã10â27 Caesium perchlorate CsClO4 3.95Ã10â3 Caesium periodate CsIO4 5.16Ã10â6 Calcium carbonate (calcite) CaCO3 3.36Ã10â9 Calcium carbonate (aragonite) CaCO3 6.0Ã10â9 Calcium fluoride CaF2 3.45Ã10â11 Calcium hydroxide Ca(OH)2 5.02Ã10â6 Calcium iodate Ca(IO3)2 6.47Ã10â6 Calcium iodate hexahydrate Ca(IO3)2·6H2O 7.10Ã10â7 Calcium molybdate CaMoO 1.46Ã10â8 Calcium oxalate monohydrate CaC2O4ÃH2O 2.32Ã10â9 Calcium phosphate Ca3(PO4)2 2.07Ã10â33 Calcium sulfate CaSO4 4.93Ã10â5 Calcium sulfate dihydrate CaSO4·2H2O 3.14Ã10â5 Calcium sulfate hemihydrate CaSO4·0.5H2O 3.1Ã10â7 Chromium(III) hydroxide Cr(OH)3 3Ã10â29 Cobalt(II) arsenate Co3(AsO4)2 6.80Ã10â29 Cobalt(II) carbonate CoCO3 1.4Ã10â13 Cobalt(II) hydroxide (blue) Co(OH)2 5.92Ã10â15 Cobalt(III) hydroxide Co(OH)3 1.6Ã10â44 Cobalt(II) iodate dihydrate Co(IO3)2·2H2O 1.21Ã10â2 Cobalt(II) phosphate Co3(PO4)2 2.05Ã10â35 Cobalt(II) sulfide CoS 4.0Ã10â21 Copper(I) bromide CuBr 6.27Ã10â9 Copper(II) carbonate CuCO3 1.4Ã10â10 Copper(I) chloride CuCl 1.72Ã10â7 Copper(I) cyanide CuCN 3.47Ã10â20 Copper(I) hydroxide CuOH 2Ã10â15 Copper(I) iodide CuI 1.27Ã10â12 Copper(I) thiocyanate CuSCN 1.77Ã10â13 Copper(II) arsenate Cu3(AsO4)2 7.95Ã10â36 Copper(II) hydroxide Cu(OH)2 1.1Ã10â15 Copper(II) iodate monohydrate Cu(IO3)2·H2O 6.94Ã10â8 Copper(II) oxalate CuC2O4 4.43Ã10â10 Copper(II) phosphate Cu3(PO4)2 1.40Ã10â37 Copper(II) sulfide CuS 6.3Ã10â26 Copper(I) sulfide Cu2S 2.5Ã10â48 Europium(III) hydroxide Eu(OH)3 9.38Ã10â27 Gallium(III) hydroxide Ga(OH)3 7.28Ã10â36 Iron(II) carbonate FeCO3 3.13Ã10â11 Iron(II) fluoride FeF2 2.36Ã10â6 Iron(II) hydroxide Fe(OH)2 4.87Ã10â17 Iron(II) sulfide FeS 1.6Ã10â19 Iron(III) hydroxide Fe(OH)3 2.79Ã10â39 Iron(III) phosphate dihydrate FePO4·2H2O 9.91Ã10â16 Lanthanum iodate La(IO3)3 7.50Ã10â12 Lead(II) bromide PbBr2 6.60Ã10â6 Lead(II) carbonate PbCO3 7.40Ã10â14 Lead(II) chloride PbCl2 1.70Ã10â5 Lead(II) chromate PbCrO4 3Ã10â13 Lead(II) fluoride PbF2 3.3Ã10â8 Lead(II) hydroxide Pb(OH)2 1.43Ã10â20 Lead(II) iodate Pb(IO3)2 3.69Ã10â13 Lead(II) iodide PbI2 9.8Ã10â9 Lead(II) oxalate PbC2O4 8.5Ã10â9 Lead(II) selenate PbSeO4 1.37Ã10â7 Lead(II) sulfate PbSO4 2.53Ã10â8 Lead(II) sulfide PbS 8.9Ã10â29 Lithium carbonate Li2CO3 8.15Ã10â4 Lithium fluoride LiF 1.84Ã10â3 Lithium phosphate Li3PO4 2.37Ã10â11 Magnesium ammonium phosphate MgNH4PO4 3Ã10â13 Magnesium carbonate MgCO3 6.82Ã10â6 Magnesium carbonate trihydrate MgCO3·3H2O 2.38Ã10â6 Magnesium carbonate pentahydrate MgCO3·5H2O 3.79Ã10â6 Magnesium fluoride MgF2 5.16Ã10â11 Magnesium hydroxide Mg(OH)2 5.61Ã10â12 Magnesium oxalate dihydrate MgC2O4·2H2O 4.83Ã10â6 Magnesium phosphate Mg3(PO4)2 1.04Ã10â24 Manganese(II) carbonate MnCO3 2.24Ã10â11 Manganese(II) iodate Mn(IO3)2 4.37Ã10â7 Manganese(II) hydroxide Mn(OH)2 2Ã10â13 Manganese(II) oxalate dihydrate MnC2O4·2H2O 1.70Ã10â7 Manganese(II) sulfide MnS 4.6Ã10â14 Mercury(I) bromide Hg2Br2 6.40Ã10â23 Mercury(I) carbonate Hg2CO3 3.6Ã10â17 Mercury(I) chloride Hg2Cl2 1.43Ã10â18 Mercury(I) fluoride Hg2F2 3.10Ã10â6 Mercury(I) iodide Hg2I2 5.2Ã10â29 Mercury(I) oxalate Hg2C2O4 1.75Ã10â13 Mercury(I) sulfate Hg2SO4 6.5Ã10â7 Mercury(I) thiocyanate Hg2(SCN)2 3.2Ã10â20 Mercury(II) bromide HgBr2 6.2Ã10â20 Mercury(II) hydroxide Hg(OH)2 3.6Ã10â26 Mercury(II) iodide HgI2 2.9Ã10â29 Mercury(II) sulfide HgS 4Ã10â53 Mercury(I) sulfide Hg2S 1.0Ã10â47 Neodymium carbonate Nd2(CO3)3 1.08Ã10â33 Nickel(II) carbonate NiCO3 1.42Ã10â7 Nickel(II) hydroxide Ni(OH)2 5.48Ã10â16 Nickel(II) iodate Ni(IO3)2 4.71Ã10â5 Nickel(II) phosphate Ni3(PO4)2 4.74Ã10â32 Nickel(II) sulfide NiS 1.1Ã10â21 Palladium(II) thiocyanate Pd(SCN)2 4.39Ã10â23 Potassium hexachloroplatinate K2PtCl6 7.48Ã10â6 Potassium perchlorate KClO4 1.05Ã10â2 Potassium periodate KIO4 3.71Ã10â4 Praseodymium hydroxide Pr(OH)3 3.39Ã10â24 Radium iodate Ra(IO3)2 1.16Ã10â9 Radium sulfate RaSO4 3.66Ã10â11 Rubidium perchlorate RbClO4 3.00Ã10â3 Scandium fluoride ScF3 5.81Ã10â24 Scandium hydroxide Sc(OH)3 2.22Ã10â31 Silver(I) acetate AgCH3COO 1.94Ã10â3 Silver(I) arsenate Ag3AsO4 1.03Ã10â22 Silver(I) bromate AgBrO3 5.38Ã10â5 Silver(I) bromide AgBr 5.35Ã10â13 Silver(I) carbonate Ag2CO3 8.46Ã10â12 Silver(I) chloride AgCl 1.77Ã10â10 Silver(I) chromate Ag2CrO4 1.12Ã10â12 Silver(I) cobalticyanide Ag3Co(CN)6 3.9Ã10â26 Silver(I) cyanide AgCN 5.97Ã10â17 Silver(I) iodate AgIO3 3.17Ã10â8 Silver(I) iodide AgI 8.52Ã10â17 Silver(I) oxalate Ag2C2O4 5.40Ã10â12 Silver(I) phosphate Ag3PO4 8.89Ã10â17 Silver(I) sulfate Ag2SO4 1.20Ã10â5 Silver(I) sulfite Ag2SO3 1.50Ã10â14 Silver(I) sulfide Ag2S 3.3Ã10â50 Silver(I) thiocyanate AgSCN 1.03Ã10â12 Strontium arsenate Sr3(AsO4)2 4.29Ã10â19 Strontium carbonate SrCO3 5.60Ã10â10 Strontium fluoride SrF2 4.33Ã10â9 Strontium iodate Sr(IO3)2 1.14Ã10â7 Strontium iodate monohydrate Sr(IO3)2·H2O 3.77Ã10â7 Strontium iodate hexahydrate Sr(IO3)2·6H2O 4.55Ã10â7 Strontium oxalate SrC2O4 5Ã10â8 Strontium phosphate Sr3(PO4)2 1Ã10â31 Strontium sulfate SrSO4 3.44Ã10â7 Thallium(I) bromate TlBrO3 1.10Ã10â4 Thallium(I) bromide TlBr 3.71Ã10â6 Thallium(I) chloride TlCl 1.86Ã10â4 Thallium(I) chromate Tl2CrO4 8.67Ã10â13 Thallium(I) hydroxide Tl(OH)3 1.68Ã10â44 Thallium(I) iodate TlIO3 3.12Ã10â6 Thallium(I) iodide TlI 5.54Ã10â8 Thallium(I) thiocyanate TlSCN 1.57Ã10â4 Thallium(I) sulfide Tl2S 6Ã10â22 Tin(II) hydroxide Sn(OH)2 5.45Ã10â27 Tin(II) sulfide SnS 3.2Ã10â28 Tin(IV) sulfide SnS2 1Ã10â70 Yttrium carbonate Y2(CO3)3 1.03Ã10â31 Yttrium fluoride YF3 8.62Ã10â21 Yttrium hydroxide Y(OH)3 1.00Ã10â22 Yttrium iodate Y(IO3)3 1.12Ã10â10 Zinc arsenate Zn3(AsO4)2 2.8Ã10â28 Zinc carbonate ZnCO3 1.46Ã10â10 Zinc carbonate monohydrate ZnCO3·H2O 5.42Ã10â11 Zinc fluoride ZnF 3.04Ã10â2 Zinc hydroxide Zn(OH)2 3Ã10â17 Zinc iodate dihydrate Zn(IO3)2·2H2O 4.1Ã10â6 Zinc oxalate dihydrate ZnC2O4·2H2O 1.38Ã10â9 Zinc selenide ZnSe 3.6Ã10â26 Zinc selenite monohydrate ZnSe·H2O 1.59Ã10â7 Zinc sulfide (alpha) ZnS 2Ã10â25 Zinc sulfide (beta) ZnS 3Ã10â23
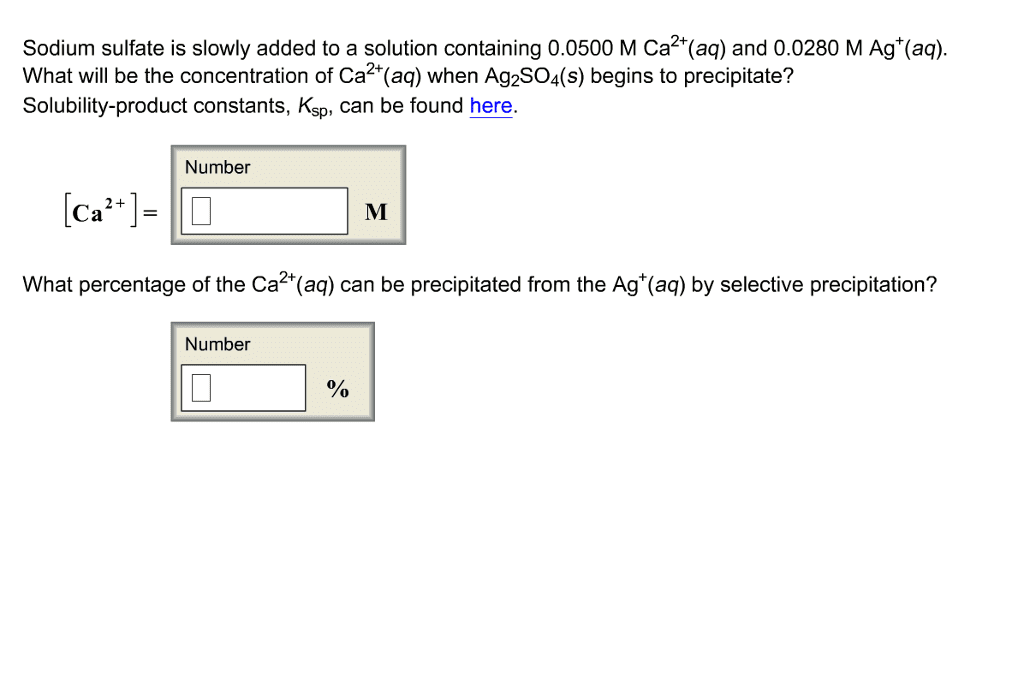
Sodium sulfate is slowly added to a solution containing 0.0500 M Ca^2+(aq) and 0.0280 M Ag^+ (aq). What will be the concentration of Ca^2+(aq) when Ag_2 SO_4 (s) begins to precipitate? Solubility-product constants, K_sp, can be found here. [Ca^2+] = What percentage of the Ca^2+ (aq) can be precipitated from the Ag^+ (aq) by selective precipitation?
Solubility-Product Constants at 298 K
| Compound | Formula | Ksp |
| Aluminium hydroxide | Al(OH)3 | 4.6Ã10â33 |
| Aluminium phosphate | AlPO4 | 9.84Ã10â21 |
| Antimony sulfide | Sb2S3 | 1.6Ã10â93 |
| Arsenic sulfide | As2S3 | 1Ã10â16 |
| Barium bromate | Ba(BrO3)2 | 2.43Ã10â4 |
| Barium carbonate | BaCO3 | 2.58Ã10â9 |
| Barium chromate | BaCrO4 | 1.17Ã10â10 |
| Barium fluoride | BaF2 | 1.84Ã10â7 |
| Barium hydroxide octahydrate | Ba(OH)2·8H2O | 2.55Ã10â4 |
| Barium iodate | Ba(IO3)2 | 4.01Ã10â9 |
| Barium iodate monohydrate | Ba(IO3)2·H2O | 1.67Ã10â9 |
| Barium molybdate | BaMoO4 | 3.54Ã10â8 |
| Barium nitrate | Ba(NO3)2 | 4.64Ã10â3 |
| Barium phosphate | Ba3(PO4)2 | 3.40Ã10â23 |
| Barium selenate | BaSeO4 | 3.40Ã10â8 |
| Barium sulfate | BaSO4 | 1.08Ã10â10 |
| Barium sulfite | BaSO3 | 5.0Ã10â10 |
| Beryllium hydroxide | Be(OH)2 | 6.92Ã10â22 |
| Bismuth arsenate | BiAsO4 | 4.43Ã10â10 |
| Bismuth iodide | BiI | 7.71Ã10â19 |
| Cadmium arsenate | Cd3(AsO4)2 | 2.2Ã10â33 |
| Cadmium carbonate | CdCO3 | 1.0Ã10â12 |
| Cadmium fluoride | CdF2 | 6.44Ã10â3 |
| Cadmium hydroxide | Cd(OH)2 | 7.2Ã10â15 |
| Cadmium iodate | Cd(IO3)2 | 2.5Ã10â8 |
| Cadmium oxalate trihydrate | CdC2O4·3H2O | 1.42Ã10â8 |
| Cadmium phosphate | Cd3(PO4)2 | 2.53Ã10â33 |
| Cadmium sulfide | CdS | 1Ã10â27 |
| Caesium perchlorate | CsClO4 | 3.95Ã10â3 |
| Caesium periodate | CsIO4 | 5.16Ã10â6 |
| Calcium carbonate (calcite) | CaCO3 | 3.36Ã10â9 |
| Calcium carbonate (aragonite) | CaCO3 | 6.0Ã10â9 |
| Calcium fluoride | CaF2 | 3.45Ã10â11 |
| Calcium hydroxide | Ca(OH)2 | 5.02Ã10â6 |
| Calcium iodate | Ca(IO3)2 | 6.47Ã10â6 |
| Calcium iodate hexahydrate | Ca(IO3)2·6H2O | 7.10Ã10â7 |
| Calcium molybdate | CaMoO | 1.46Ã10â8 |
| Calcium oxalate monohydrate | CaC2O4ÃH2O | 2.32Ã10â9 |
| Calcium phosphate | Ca3(PO4)2 | 2.07Ã10â33 |
| Calcium sulfate | CaSO4 | 4.93Ã10â5 |
| Calcium sulfate dihydrate | CaSO4·2H2O | 3.14Ã10â5 |
| Calcium sulfate hemihydrate | CaSO4·0.5H2O | 3.1Ã10â7 |
| Chromium(III) hydroxide | Cr(OH)3 | 3Ã10â29 |
| Cobalt(II) arsenate | Co3(AsO4)2 | 6.80Ã10â29 |
| Cobalt(II) carbonate | CoCO3 | 1.4Ã10â13 |
| Cobalt(II) hydroxide (blue) | Co(OH)2 | 5.92Ã10â15 |
| Cobalt(III) hydroxide | Co(OH)3 | 1.6Ã10â44 |
| Cobalt(II) iodate dihydrate | Co(IO3)2·2H2O | 1.21Ã10â2 |
| Cobalt(II) phosphate | Co3(PO4)2 | 2.05Ã10â35 |
| Cobalt(II) sulfide | CoS | 4.0Ã10â21 |
| Copper(I) bromide | CuBr | 6.27Ã10â9 |
| Copper(II) carbonate | CuCO3 | 1.4Ã10â10 |
| Copper(I) chloride | CuCl | 1.72Ã10â7 |
| Copper(I) cyanide | CuCN | 3.47Ã10â20 |
| Copper(I) hydroxide | CuOH | 2Ã10â15 |
| Copper(I) iodide | CuI | 1.27Ã10â12 |
| Copper(I) thiocyanate | CuSCN | 1.77Ã10â13 |
| Copper(II) arsenate | Cu3(AsO4)2 | 7.95Ã10â36 |
| Copper(II) hydroxide | Cu(OH)2 | 1.1Ã10â15 |
| Copper(II) iodate monohydrate | Cu(IO3)2·H2O | 6.94Ã10â8 |
| Copper(II) oxalate | CuC2O4 | 4.43Ã10â10 |
| Copper(II) phosphate | Cu3(PO4)2 | 1.40Ã10â37 |
| Copper(II) sulfide | CuS | 6.3Ã10â26 |
| Copper(I) sulfide | Cu2S | 2.5Ã10â48 |
| Europium(III) hydroxide | Eu(OH)3 | 9.38Ã10â27 |
| Gallium(III) hydroxide | Ga(OH)3 | 7.28Ã10â36 |
| Iron(II) carbonate | FeCO3 | 3.13Ã10â11 |
| Iron(II) fluoride | FeF2 | 2.36Ã10â6 |
| Iron(II) hydroxide | Fe(OH)2 | 4.87Ã10â17 |
| Iron(II) sulfide | FeS | 1.6Ã10â19 |
| Iron(III) hydroxide | Fe(OH)3 | 2.79Ã10â39 |
| Iron(III) phosphate dihydrate | FePO4·2H2O | 9.91Ã10â16 |
| Lanthanum iodate | La(IO3)3 | 7.50Ã10â12 |
| Lead(II) bromide | PbBr2 | 6.60Ã10â6 |
| Lead(II) carbonate | PbCO3 | 7.40Ã10â14 |
| Lead(II) chloride | PbCl2 | 1.70Ã10â5 |
| Lead(II) chromate | PbCrO4 | 3Ã10â13 |
| Lead(II) fluoride | PbF2 | 3.3Ã10â8 |
| Lead(II) hydroxide | Pb(OH)2 | 1.43Ã10â20 |
| Lead(II) iodate | Pb(IO3)2 | 3.69Ã10â13 |
| Lead(II) iodide | PbI2 | 9.8Ã10â9 |
| Lead(II) oxalate | PbC2O4 | 8.5Ã10â9 |
| Lead(II) selenate | PbSeO4 | 1.37Ã10â7 |
| Lead(II) sulfate | PbSO4 | 2.53Ã10â8 |
| Lead(II) sulfide | PbS | 8.9Ã10â29 |
| Lithium carbonate | Li2CO3 | 8.15Ã10â4 |
| Lithium fluoride | LiF | 1.84Ã10â3 |
| Lithium phosphate | Li3PO4 | 2.37Ã10â11 |
| Magnesium ammonium phosphate | MgNH4PO4 | 3Ã10â13 |
| Magnesium carbonate | MgCO3 | 6.82Ã10â6 |
| Magnesium carbonate trihydrate | MgCO3·3H2O | 2.38Ã10â6 |
| Magnesium carbonate pentahydrate | MgCO3·5H2O | 3.79Ã10â6 |
| Magnesium fluoride | MgF2 | 5.16Ã10â11 |
| Magnesium hydroxide | Mg(OH)2 | 5.61Ã10â12 |
| Magnesium oxalate dihydrate | MgC2O4·2H2O | 4.83Ã10â6 |
| Magnesium phosphate | Mg3(PO4)2 | 1.04Ã10â24 |
| Manganese(II) carbonate | MnCO3 | 2.24Ã10â11 |
| Manganese(II) iodate | Mn(IO3)2 | 4.37Ã10â7 |
| Manganese(II) hydroxide | Mn(OH)2 | 2Ã10â13 |
| Manganese(II) oxalate dihydrate | MnC2O4·2H2O | 1.70Ã10â7 |
| Manganese(II) sulfide | MnS | 4.6Ã10â14 |
| Mercury(I) bromide | Hg2Br2 | 6.40Ã10â23 |
| Mercury(I) carbonate | Hg2CO3 | 3.6Ã10â17 |
| Mercury(I) chloride | Hg2Cl2 | 1.43Ã10â18 |
| Mercury(I) fluoride | Hg2F2 | 3.10Ã10â6 |
| Mercury(I) iodide | Hg2I2 | 5.2Ã10â29 |
| Mercury(I) oxalate | Hg2C2O4 | 1.75Ã10â13 |
| Mercury(I) sulfate | Hg2SO4 | 6.5Ã10â7 |
| Mercury(I) thiocyanate | Hg2(SCN)2 | 3.2Ã10â20 |
| Mercury(II) bromide | HgBr2 | 6.2Ã10â20 |
| Mercury(II) hydroxide | Hg(OH)2 | 3.6Ã10â26 |
| Mercury(II) iodide | HgI2 | 2.9Ã10â29 |
| Mercury(II) sulfide | HgS | 4Ã10â53 |
| Mercury(I) sulfide | Hg2S | 1.0Ã10â47 |
| Neodymium carbonate | Nd2(CO3)3 | 1.08Ã10â33 |
| Nickel(II) carbonate | NiCO3 | 1.42Ã10â7 |
| Nickel(II) hydroxide | Ni(OH)2 | 5.48Ã10â16 |
| Nickel(II) iodate | Ni(IO3)2 | 4.71Ã10â5 |
| Nickel(II) phosphate | Ni3(PO4)2 | 4.74Ã10â32 |
| Nickel(II) sulfide | NiS | 1.1Ã10â21 |
| Palladium(II) thiocyanate | Pd(SCN)2 | 4.39Ã10â23 |
| Potassium hexachloroplatinate | K2PtCl6 | 7.48Ã10â6 |
| Potassium perchlorate | KClO4 | 1.05Ã10â2 |
| Potassium periodate | KIO4 | 3.71Ã10â4 |
| Praseodymium hydroxide | Pr(OH)3 | 3.39Ã10â24 |
| Radium iodate | Ra(IO3)2 | 1.16Ã10â9 |
| Radium sulfate | RaSO4 | 3.66Ã10â11 |
| Rubidium perchlorate | RbClO4 | 3.00Ã10â3 |
| Scandium fluoride | ScF3 | 5.81Ã10â24 |
| Scandium hydroxide | Sc(OH)3 | 2.22Ã10â31 |
| Silver(I) acetate | AgCH3COO | 1.94Ã10â3 |
| Silver(I) arsenate | Ag3AsO4 | 1.03Ã10â22 |
| Silver(I) bromate | AgBrO3 | 5.38Ã10â5 |
| Silver(I) bromide | AgBr | 5.35Ã10â13 |
| Silver(I) carbonate | Ag2CO3 | 8.46Ã10â12 |
| Silver(I) chloride | AgCl | 1.77Ã10â10 |
| Silver(I) chromate | Ag2CrO4 | 1.12Ã10â12 |
| Silver(I) cobalticyanide | Ag3Co(CN)6 | 3.9Ã10â26 |
| Silver(I) cyanide | AgCN | 5.97Ã10â17 |
| Silver(I) iodate | AgIO3 | 3.17Ã10â8 |
| Silver(I) iodide | AgI | 8.52Ã10â17 |
| Silver(I) oxalate | Ag2C2O4 | 5.40Ã10â12 |
| Silver(I) phosphate | Ag3PO4 | 8.89Ã10â17 |
| Silver(I) sulfate | Ag2SO4 | 1.20Ã10â5 |
| Silver(I) sulfite | Ag2SO3 | 1.50Ã10â14 |
| Silver(I) sulfide | Ag2S | 3.3Ã10â50 |
| Silver(I) thiocyanate | AgSCN | 1.03Ã10â12 |
| Strontium arsenate | Sr3(AsO4)2 | 4.29Ã10â19 |
| Strontium carbonate | SrCO3 | 5.60Ã10â10 |
| Strontium fluoride | SrF2 | 4.33Ã10â9 |
| Strontium iodate | Sr(IO3)2 | 1.14Ã10â7 |
| Strontium iodate monohydrate | Sr(IO3)2·H2O | 3.77Ã10â7 |
| Strontium iodate hexahydrate | Sr(IO3)2·6H2O | 4.55Ã10â7 |
| Strontium oxalate | SrC2O4 | 5Ã10â8 |
| Strontium phosphate | Sr3(PO4)2 | 1Ã10â31 |
| Strontium sulfate | SrSO4 | 3.44Ã10â7 |
| Thallium(I) bromate | TlBrO3 | 1.10Ã10â4 |
| Thallium(I) bromide | TlBr | 3.71Ã10â6 |
| Thallium(I) chloride | TlCl | 1.86Ã10â4 |
| Thallium(I) chromate | Tl2CrO4 | 8.67Ã10â13 |
| Thallium(I) hydroxide | Tl(OH)3 | 1.68Ã10â44 |
| Thallium(I) iodate | TlIO3 | 3.12Ã10â6 |
| Thallium(I) iodide | TlI | 5.54Ã10â8 |
| Thallium(I) thiocyanate | TlSCN | 1.57Ã10â4 |
| Thallium(I) sulfide | Tl2S | 6Ã10â22 |
| Tin(II) hydroxide | Sn(OH)2 | 5.45Ã10â27 |
| Tin(II) sulfide | SnS | 3.2Ã10â28 |
| Tin(IV) sulfide | SnS2 | 1Ã10â70 |
| Yttrium carbonate | Y2(CO3)3 | 1.03Ã10â31 |
| Yttrium fluoride | YF3 | 8.62Ã10â21 |
| Yttrium hydroxide | Y(OH)3 | 1.00Ã10â22 |
| Yttrium iodate | Y(IO3)3 | 1.12Ã10â10 |
| Zinc arsenate | Zn3(AsO4)2 | 2.8Ã10â28 |
| Zinc carbonate | ZnCO3 | 1.46Ã10â10 |
| Zinc carbonate monohydrate | ZnCO3·H2O | 5.42Ã10â11 |
| Zinc fluoride | ZnF | 3.04Ã10â2 |
| Zinc hydroxide | Zn(OH)2 | 3Ã10â17 |
| Zinc iodate dihydrate | Zn(IO3)2·2H2O | 4.1Ã10â6 |
| Zinc oxalate dihydrate | ZnC2O4·2H2O | 1.38Ã10â9 |
| Zinc selenide | ZnSe | 3.6Ã10â26 |
| Zinc selenite monohydrate | ZnSe·H2O | 1.59Ã10â7 |
| Zinc sulfide (alpha) | ZnS | 2Ã10â25 |
| Zinc sulfide (beta) | ZnS | 3Ã10â23 |

Sodium sulfate is slowly added to a solution containing 0.0500 M Ca^2+(aq) and 0.0280 M Ag^+ (aq). What will be the concentration of Ca^2+(aq) when Ag_2 SO_4 (s) begins to precipitate? Solubility-product constants, K_sp, can be found here. [Ca^2+] = What percentage of the Ca^2+ (aq) can be precipitated from the Ag^+ (aq) by selective precipitation?
Beverley SmithLv2
18 Apr 2019
