1
answer
0
watching
260
views
11 Nov 2019
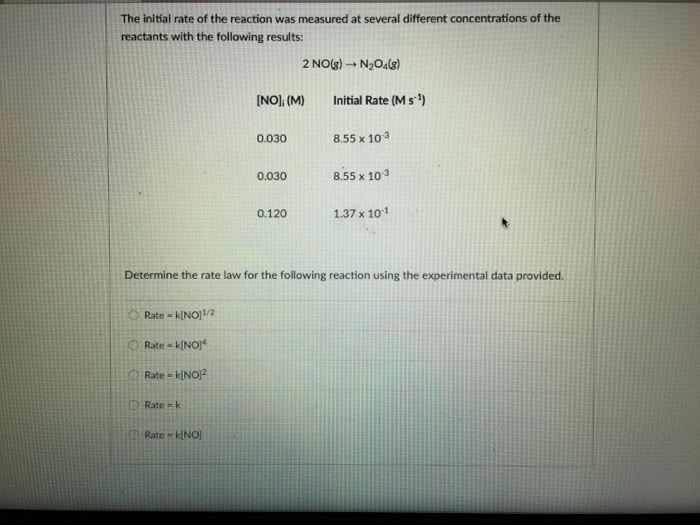
The initial rate of the reaction was measured at several different concentrations of the reactants with the following results:
2 NO(g) â N2O4(g)
[NO]i (M)
Initial Rate (M s-1)
0.030
8.55 x 10-3
0.030
8.55 x 10-3
0.120
1.37 x 10-1
Determine the rate law for the following reaction using the experimental data provided.
A) Rate = k[NO]1/2
B) Rate = k[NO]2
C)Rate = k[NO]4
D) Rate = k
E)Rate = k[NO]
I have no idea how to even approach this :(
this was all the information that was given
The initial rate of the reaction was measured at several different concentrations of the reactants with the following results:
2 NO(g) â N2O4(g)
| [NO]i (M) | Initial Rate (M s-1) |
| 0.030 | 8.55 x 10-3 |
| 0.030 | 8.55 x 10-3 |
| 0.120 | 1.37 x 10-1 |
Determine the rate law for the following reaction using the experimental data provided.
A) Rate = k[NO]1/2
B) Rate = k[NO]2
C)Rate = k[NO]4
D) Rate = k
E)Rate = k[NO]
I have no idea how to even approach this :(
this was all the information that was given
Beverley SmithLv2
6 Jun 2019