1
answer
0
watching
105
views
11 Nov 2019


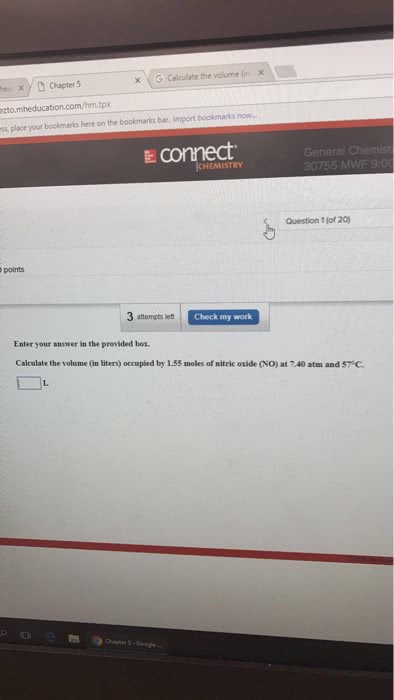
place your bookmarks here on the bookmarks bar. Import bookmarks now.. è£connect CHEMISTRY Question 5 (of 20) ints 3 attempts let Check my work Be sure to answer all parts A sample of natural gas contains 6.501 moles of methane (CHa). 0.954 moles of ethane (C,H) and 0.310 moles of propane (CHg). If the total pressure of the gases is 2.77 atm, what are the partial pressures of the gases? PCHat oe
1
answer
0
watching
105
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Trinidad TremblayLv2
10 Jun 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232