1
answer
0
watching
177
views
11 Nov 2019
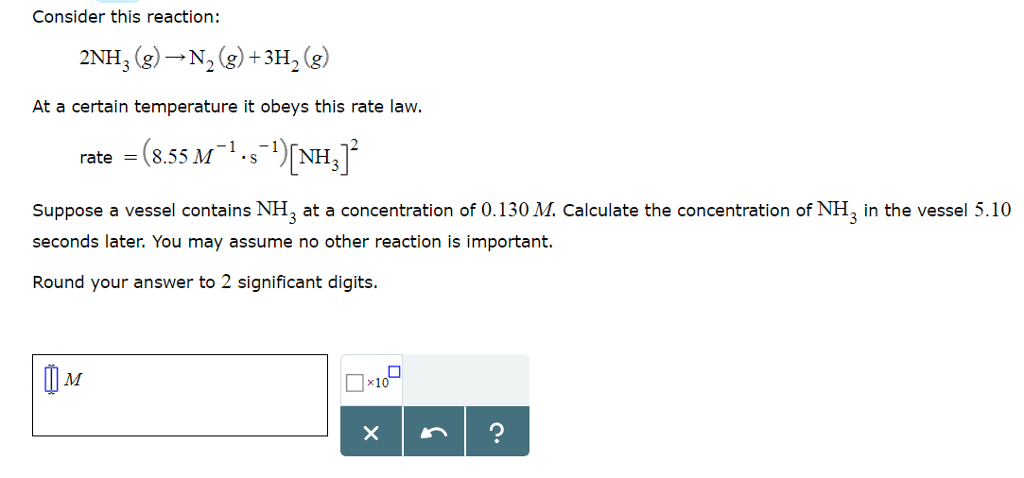
Consider this reaction: 2NH3=N2 =3H2
At a certain temperature it obeys this rate law: rate = (0.757 M^-1 S^-1) [NH3]^2
Suppose a vessel contains NH3 at a concentration of 1.12M. Calculate how long it takes for the concentration to decrease to 0.179M. You may assume no other reaction is important. Round your answer to 2 Sig. Figs.
Consider this reaction: 2NH3=N2 =3H2
At a certain temperature it obeys this rate law: rate = (0.757 M^-1 S^-1) [NH3]^2
Suppose a vessel contains NH3 at a concentration of 1.12M. Calculate how long it takes for the concentration to decrease to 0.179M. You may assume no other reaction is important. Round your answer to 2 Sig. Figs.
Keith LeannonLv2
29 Mar 2019