1
answer
0
watching
275
views
11 Nov 2019
For the instructions page, disregard what it says about the Microsoft Word report form and just include in your answer what it says after that! Thanks!

For the instructions page, disregard what it says about the Microsoft Word report form and just include in your answer what it says after that! Thanks!






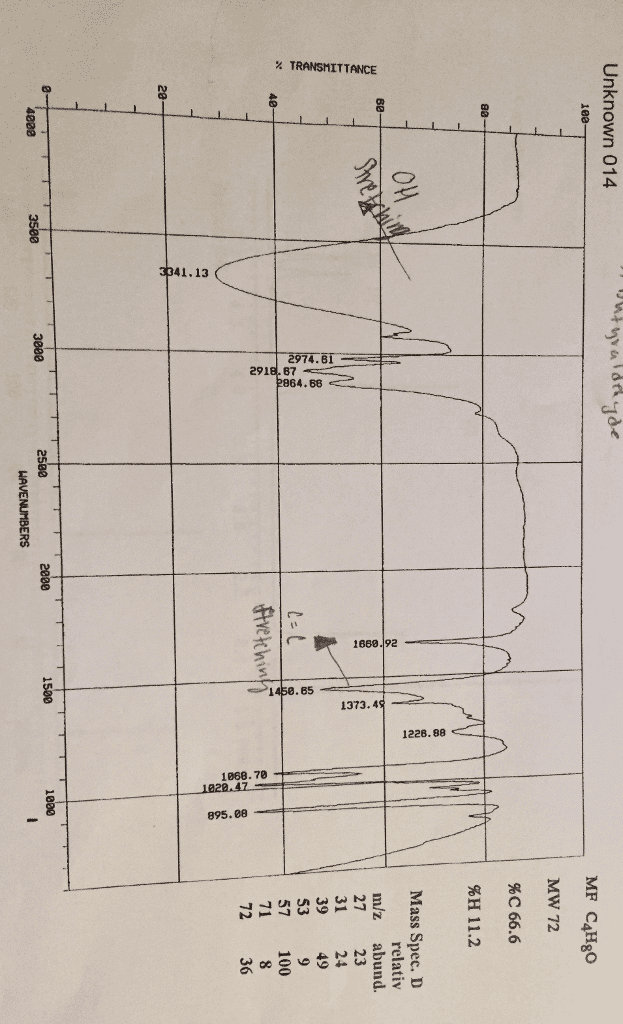
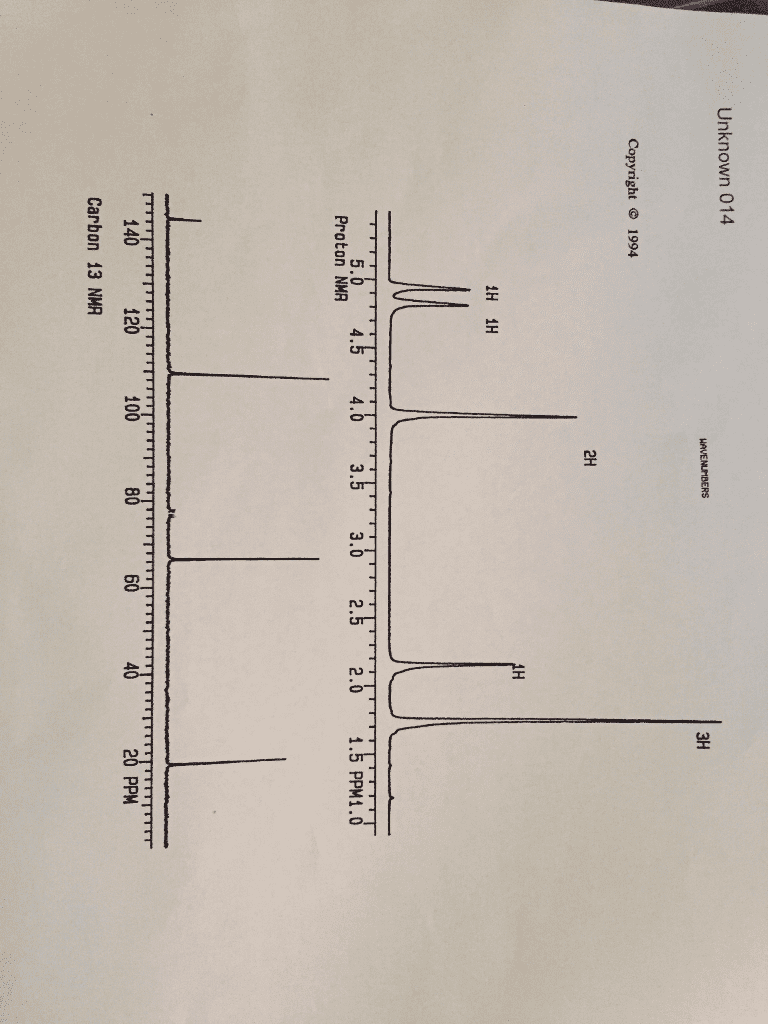
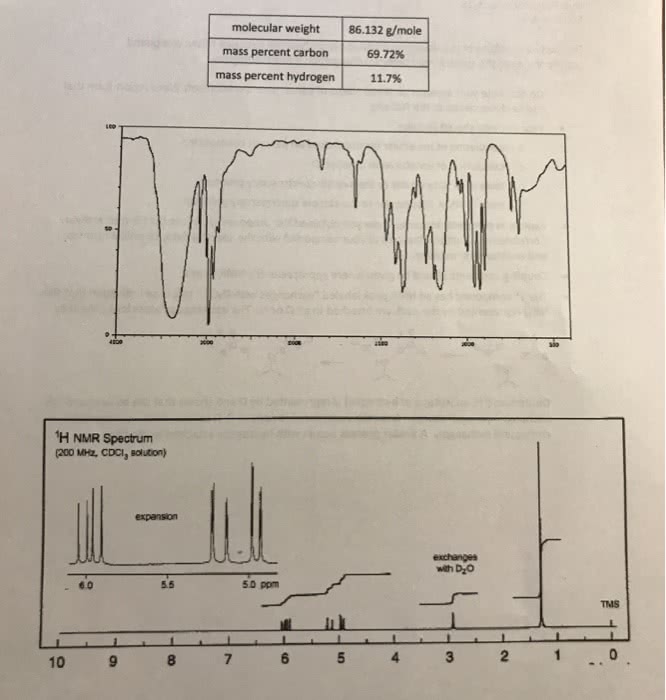
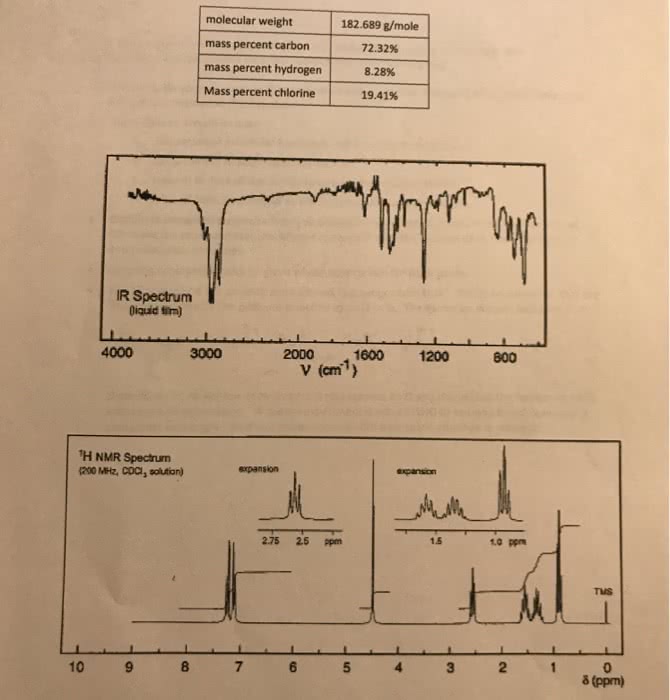
For the two problems of this assignment you are to deduce the structure of the compound corresponding to the spectra and chemical composition information given. Do not write your answers on these pieces of paper. Use the Microsoft Word report form that can be downloaded at the D2L site. . Your answers should include: o o o o calculation of molecular formula based on percent composition calculation of unsaturation number, Ω table of IR data similar to the sample spectroscopy problem table of NMR data similar to the sample spectroscopy problem Explain in complete sentences how you obtained the proposed structure that is your answer. Correlate the structural features of your compound with the spectral data, chemical formula and unsaturation number. Coupling constants should be given where appropriate for NMR peaks. 'H(s) represented by the peak are attached to an O or N. The exchange process looks like this: The 1t compound has an NMR peak labeled "exchanges with D,O". This is an indication that the OH Deuterium (H, an isotope of hydrogen) is represented by D and shows that the solvent and OH exchange hydrogen atoms. A special experiment is done in D10 to see which hydrogen(s) in a compound exchanges. A similar process occurs with hydrogens attached to nitrogen.
Casey DurganLv2
19 Jun 2019