1
answer
0
watching
123
views
11 Nov 2019
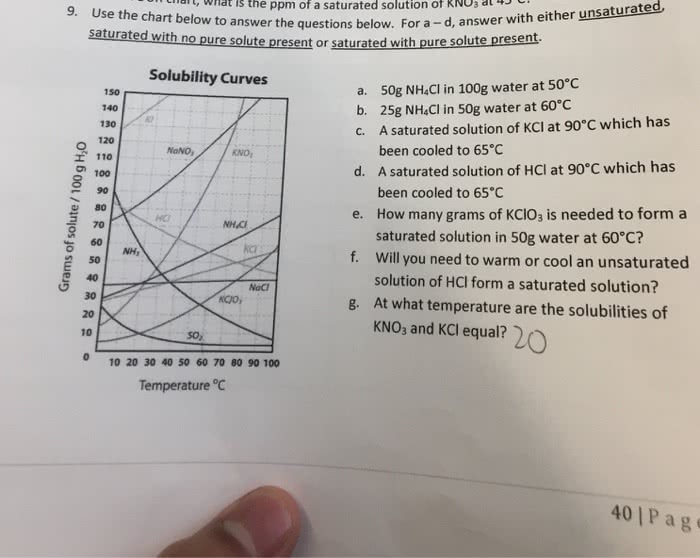
Part A: In lab, 74.42 grams of H2O is saturated with KNO3 at 75.0ºC where the solubility is 145 g KNO3/100 g H2O. It is then cooled to 20.0ºC where the solubility is 35g KNO3/100 g H2O. How many grams of KNO3 will precipitate out of the solution? Put your answer in 3 significant figures.
âPart B: What is the molarity of a solution that has 55.78 g KNO3 dissolved in 100 g of water at 45ºC and a solution density of 1.23 g/ml. Put your answer in 3 significant figures.
Part A: In lab, 74.42 grams of H2O is saturated with KNO3 at 75.0ºC where the solubility is 145 g KNO3/100 g H2O. It is then cooled to 20.0ºC where the solubility is 35g KNO3/100 g H2O. How many grams of KNO3 will precipitate out of the solution? Put your answer in 3 significant figures.
âPart B: What is the molarity of a solution that has 55.78 g KNO3 dissolved in 100 g of water at 45ºC and a solution density of 1.23 g/ml. Put your answer in 3 significant figures.
Casey DurganLv2
28 Sep 2019