1
answer
0
watching
1,241
views
11 Nov 2019
Calculate the percent yield , determined melting point and show reaction mechanism pleas


Calculate the percent yield , determined melting point and show reaction mechanism pleas




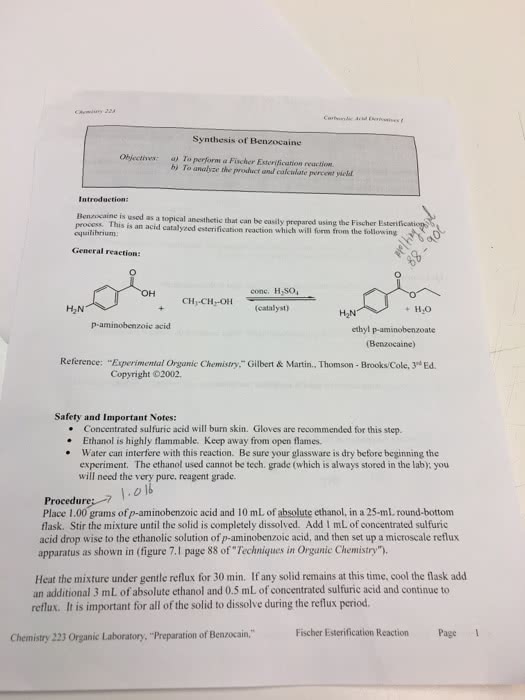
Synthesis of Benzocaine Obfectives:aToperform a Fischer Esterification reaction bj To analyze the product and calculate percent viclad Introduction: Benzocaine is used as a topical anesthctic that can be easily prepared using the Fischer E process. This is an acid catalyzed esterification reaction which will form from the following equilibrium General reaction: conc. H,SO H2N (eatalyst) ethyl p-aminobenzoate Benzocaine) Reference: "Experimental Organic Chemistry."Gilbert &Martin., Thomson - Brooks/Cole, 3* Ed. Copyright ©2002. Safety and Important Notes: . Concentrated sulfuric acid will burn skin. Gloves are recommended for this step. e Ethanol is highly flammable. Keep away from open flames. . Water can interfere with this reaction. Be sure your glassware is dry before beginning the experiment. The ethanol used cannot be tech. grade (which is always stored in the lab); you will need the very pure, reagent grade. Procedure: Place 1.00 grams of p-aminobenzoic acid and 10 mL of absolute ethanol, in a 25-mL round-bottom flask. Stir the mixture until the solid is completely dissolved. Add 1 mL of concentrated sulfuric acid drop wise to the ethanolic solution of p-aminobenzoic acid, and then set up a microscale reflux apparatus as shown in (figure 7.1 page 88 of Techniques in Organic Chemistry"). Heat the mixture under gentle reflux for 30 min. If any solid remains at this time, cool the flask add an additional 3 mL of absolute ethanol and 0.5 mL of concentrated sulfuric acid and continue to reflux. It is important for all of the solid to dissolve during the reflux period. Fischer Esterification Reaction Page Chemistry 223 Organic Laboratory, "Preparation of Benzocain,"
Jamar FerryLv2
9 Mar 2019



