1
answer
1
watching
823
views
11 Nov 2019
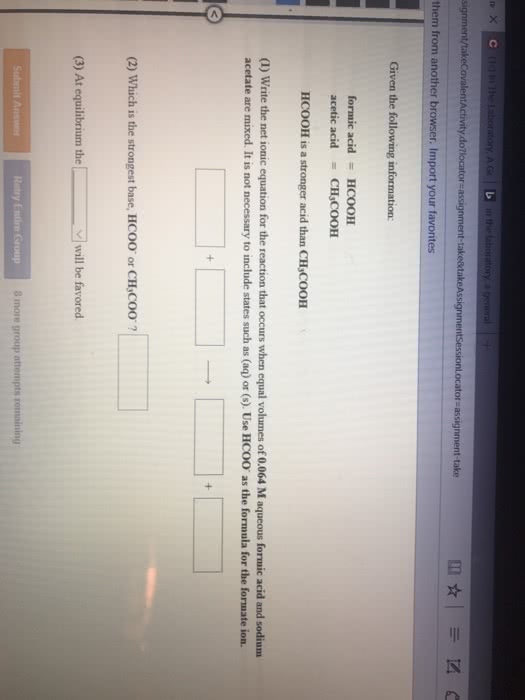
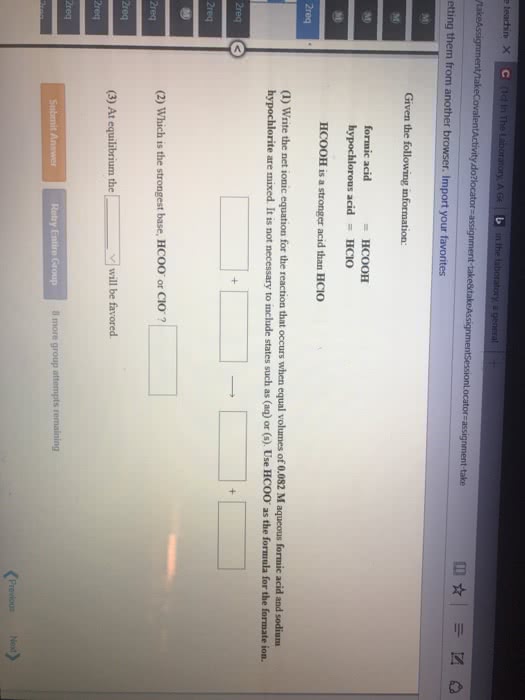
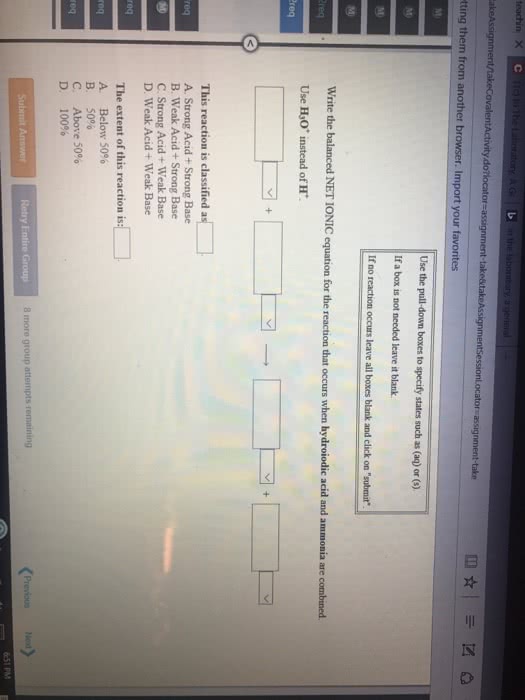
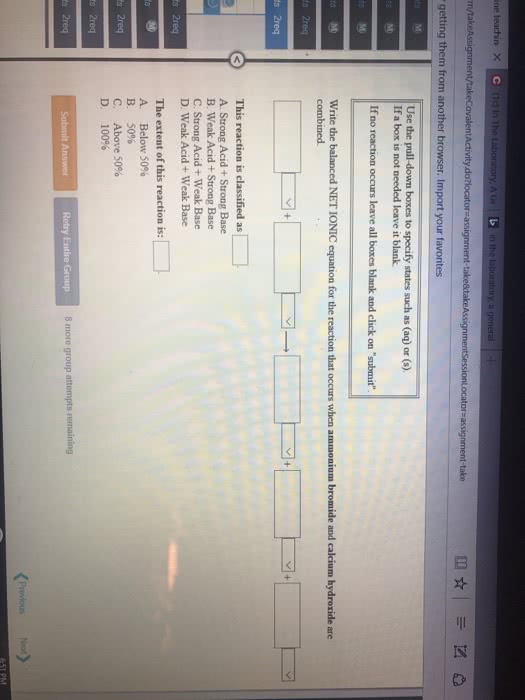
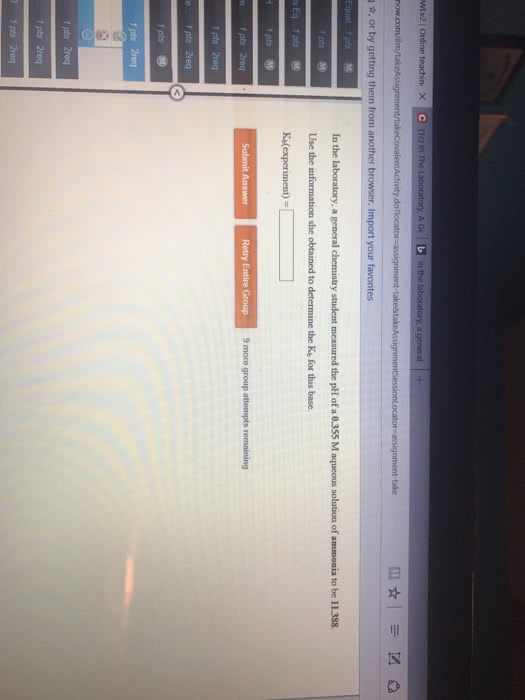
Given the following information:
formic acid HCOOH Ka = 1.8Ã10-4 hydrocyanic acid HCN Ka = 4.0Ã10-10
(1) Write the net ionic equation for the reaction that occurs when equal volumes of 0.326 Maqueous formic acid and sodium cyanide are mixed. It is not necessary to include states such as (aq) or (s). Use HCOO- as the formula for the formate ion.
(2) At equilibrium will the reactants or products be favored?
(3) Will the pH of the resulting solution be less than seven, greater than seven or equal to seven?
Given the following information:
| formic acid | HCOOH | Ka = 1.8Ã10-4 |
| hydrocyanic acid | HCN | Ka = 4.0Ã10-10 |
(1) Write the net ionic equation for the reaction that occurs when equal volumes of 0.326 Maqueous formic acid and sodium cyanide are mixed. It is not necessary to include states such as (aq) or (s). Use HCOO- as the formula for the formate ion.
(2) At equilibrium will the reactants or products be favored?
(3) Will the pH of the resulting solution be less than seven, greater than seven or equal to seven?
Hubert KochLv2
11 Nov 2019