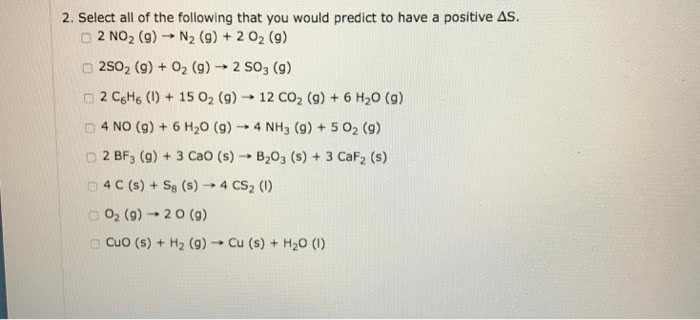
1
answer
0
watching
717
views
rubywolf159Lv1
28 Sep 2019
4. Predict the sign of the entropy change for the following processes.
a. Ag+(aq) + Cl? (aq) ? AgCl (s)
b. 2 Mg (s) + O2 (g) ? 2 MgO (s)
c. 4 NH3 (g) + 5 O2 (g) ? 4 NO (g) + 6 H2O (l)
d. 3 NO2 (g) + H2O (l) ? 2 HNO3 (aq) + NO (g)
5. Calculate ?S298 ° for the following changes.
(a) SnCl4(l) ? SnCl4(g)
(b) CS2(g) ? CS2(l)
6. Calculate ?S298 ° for the following changes.
(a) CaO (s) + H2O (l) ? Ca(OH)2 (s)
(b) Fe2O3 (s) + 3 CO (g) ? 2 Fe (s) + 3 CO2 (g)
(c) CS2 (g) + 3 Cl2 (g) ? CCl4 (g) + S2Cl2 (g)
(d) 2 Al (s) + Fe2O3 (s) ? Al2O3 (s) + 2 Fe (s)
4. Predict the sign of the entropy change for the following processes.
a. Ag+(aq) + Cl? (aq) ? AgCl (s)
b. 2 Mg (s) + O2 (g) ? 2 MgO (s)
c. 4 NH3 (g) + 5 O2 (g) ? 4 NO (g) + 6 H2O (l)
d. 3 NO2 (g) + H2O (l) ? 2 HNO3 (aq) + NO (g)
5. Calculate ?S298 ° for the following changes.
(a) SnCl4(l) ? SnCl4(g)
(b) CS2(g) ? CS2(l)
6. Calculate ?S298 ° for the following changes.
(a) CaO (s) + H2O (l) ? Ca(OH)2 (s)
(b) Fe2O3 (s) + 3 CO (g) ? 2 Fe (s) + 3 CO2 (g)
(c) CS2 (g) + 3 Cl2 (g) ? CCl4 (g) + S2Cl2 (g)
(d) 2 Al (s) + Fe2O3 (s) ? Al2O3 (s) + 2 Fe (s)
Liked by bwb4luo
Casey DurganLv2
28 Sep 2019