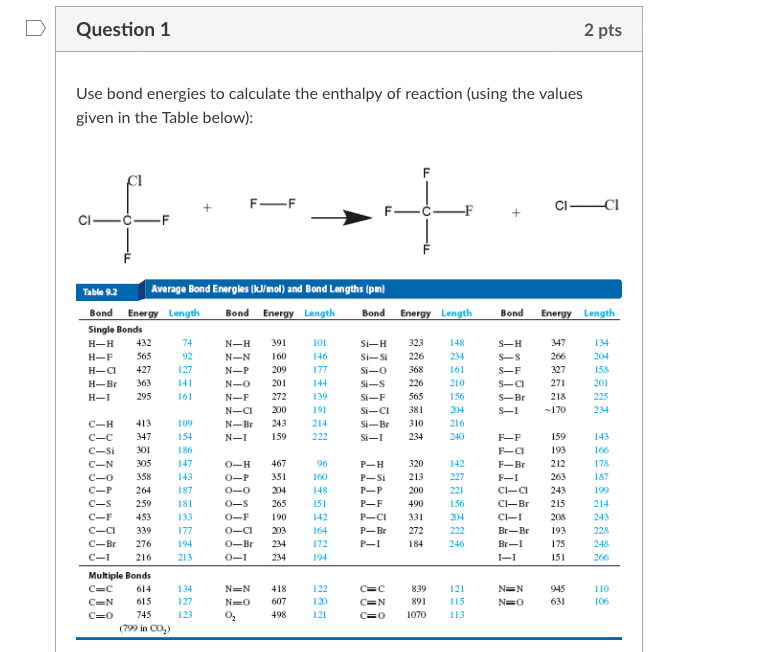
1
answer
0
watching
434
views
11 Nov 2019
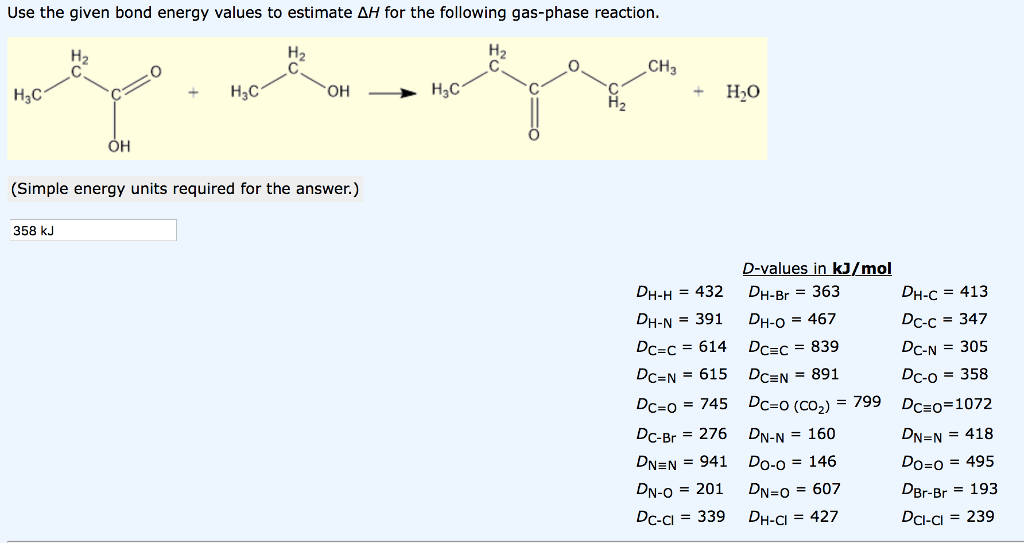
Use the given bond energy values to estimate AH for the following gas-phase reaction CH3 H3C + H3C H3C + HO OH (Simple energy units required for the answer.) 358 kJ DH-H=432 DH-N-391 DC-C = 614 DCzN-615 DC-o-745 DC-Br=276 DNEN=941 DN-0= 201 DC-cl=339 values in kJ/mol DH-Br=363 DH-0 = 467 DC=C = 839 DC-N-891 DC-0 (CO2,-799 DN-N=160 Do-o=146 DN=0= 607 DH-Cl=427 DH-C = 413 Dc-c - 347 DC-N = 305 DC-o = 358 DCz0-1072 DN=N = 418 Do-o = 495 Br-Br = 193 DCI-CI = 239

Use the given bond energy values to estimate AH for the following gas-phase reaction CH3 H3C + H3C H3C + HO OH (Simple energy units required for the answer.) 358 kJ DH-H=432 DH-N-391 DC-C = 614 DCzN-615 DC-o-745 DC-Br=276 DNEN=941 DN-0= 201 DC-cl=339 values in kJ/mol DH-Br=363 DH-0 = 467 DC=C = 839 DC-N-891 DC-0 (CO2,-799 DN-N=160 Do-o=146 DN=0= 607 DH-Cl=427 DH-C = 413 Dc-c - 347 DC-N = 305 DC-o = 358 DCz0-1072 DN=N = 418 Do-o = 495 Br-Br = 193 DCI-CI = 239
Hubert KochLv2
28 Oct 2019