0
answers
0
watching
622
views
11 Nov 2019
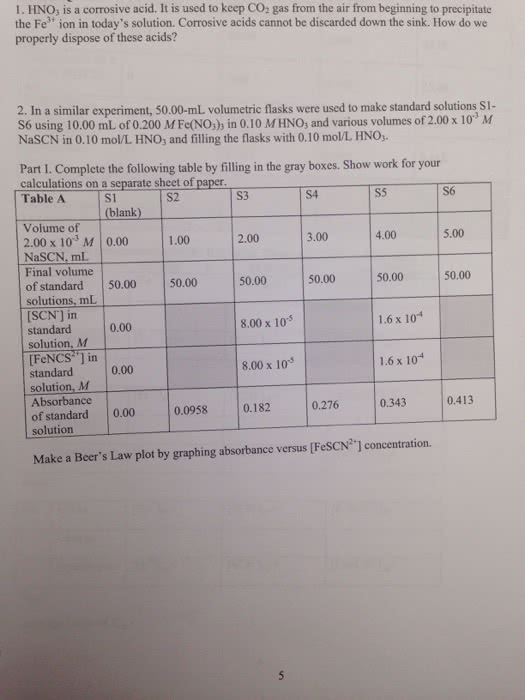
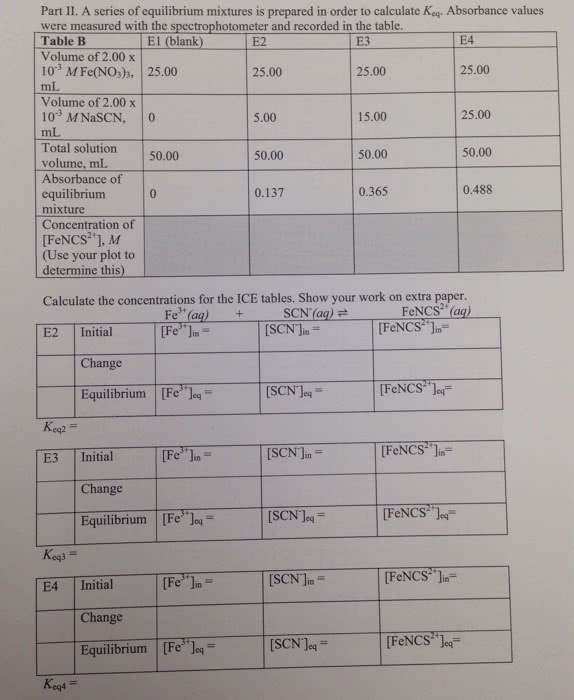
I need help to calculate the equilibrium of FeNCS2+ ion concentration

1. Preparing Standard Solutions for the Beer's Law Plot concentration of Fe(NO3)3 in 1.0 x 10-1M HNO, solution, mol L--| - 0-2 0.2 concentration of NasCN in 1.0x10-'M HNO3 solution, mol L-1 O.S flask number S1 S1 S2 S3 S4 S5 S6 S4 S5 S6 (blank) S2 S3 volume of NasCN in 1.0x10-1M HNO3 solution added, mL 0-00 1.00 2.00 3.00 4.00 5.00 initial son lon concentration, 0 0.01 0.02 0.03 0.01 0.06 mol L-1 equilibrium Fencsâ ion 0 - - - - - equilibrium FeNCS2+ ion concentration, mol L-1
I need help to calculate the equilibrium of FeNCS2+ ion concentration

1. Preparing Standard Solutions for the Beer's Law Plot concentration of Fe(NO3)3 in 1.0 x 10-1M HNO, solution, mol L--| - 0-2 0.2 concentration of NasCN in 1.0x10-'M HNO3 solution, mol L-1 O.S flask number S1 S1 S2 S3 S4 S5 S6 S4 S5 S6 (blank) S2 S3 volume of NasCN in 1.0x10-1M HNO3 solution added, mL 0-00 1.00 2.00 3.00 4.00 5.00 initial son lon concentration, 0 0.01 0.02 0.03 0.01 0.06 mol L-1 equilibrium Fencsâ ion 0 - - - - - equilibrium FeNCS2+ ion concentration, mol L-1