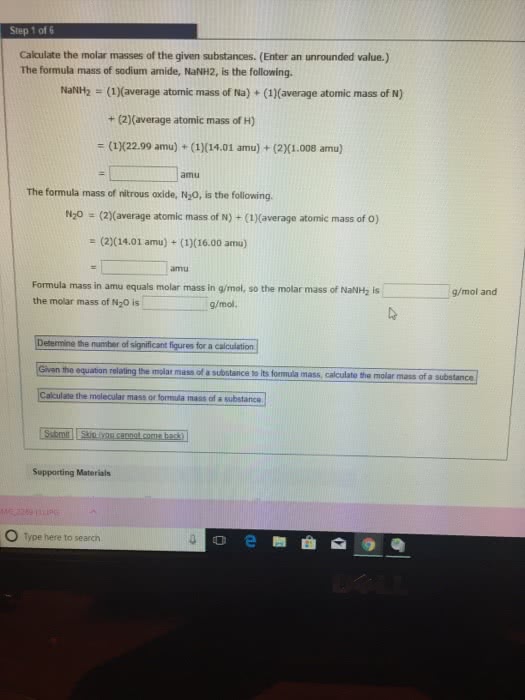
1
answer
0
watching
343
views
11 Nov 2019
The molar mass of KCl is: (use atomic weights to whole numbers). A.74 g. B.74 amu. C.none of these
The molar mass of KCl is: (use atomic weights to whole numbers). A.74 g. B.74 amu. C.none of these
1
answer
0
watching
343
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Jamar FerryLv2
15 Sep 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232
Related questions
Calculate the missing values in the table if the average atomic mass of germanium is .
| Isotope | Isotope Mass ( |
Percent Abundance ( |
| 69.9243 | 20.50 | |
| - | 27.40 | |
| 72.9235 | 7.80 | |
| 73.9212 | - | |
| 75.9214 | 7.80 |
The formula weight for Mg(OH)2 is: (use atomic weights to the whole number)
| A. | 41 amu | |
| B. | 42 amu | |
| C. | 59 amu |