1
answer
0
watching
441
views
11 Nov 2019
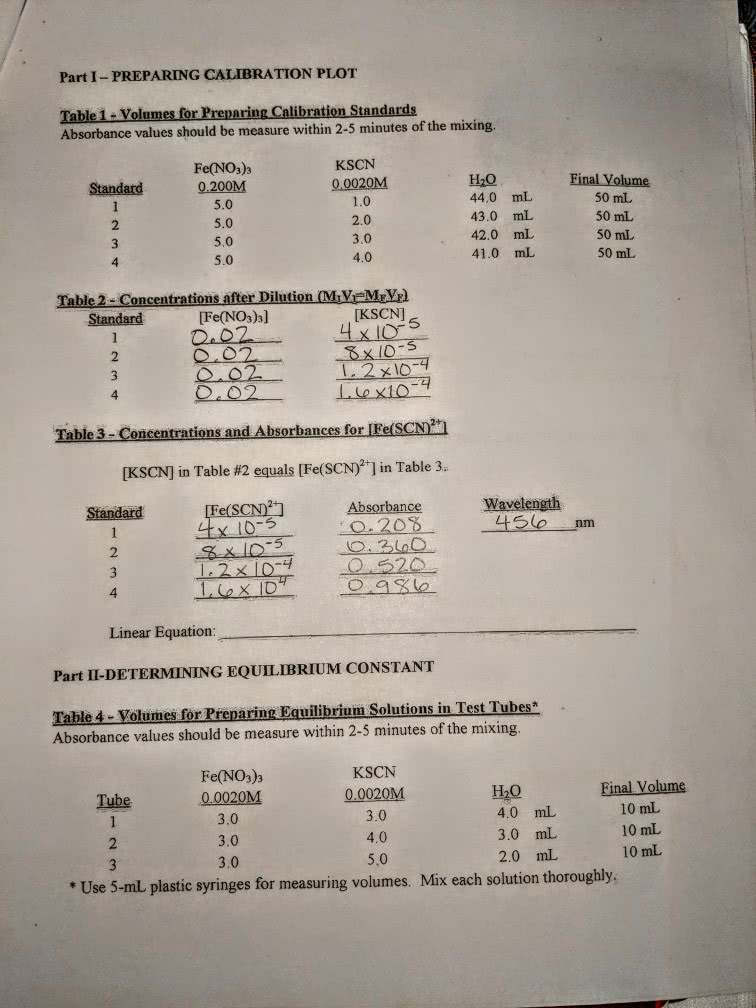
Find the concentration of Fe(SCN)2+ for each test tube.
30mL of 0.200M Fe(NO3)3 was prepared. Then, 15mL of 0.0020M KSCN was prepraed in another beaker. 10.0mL of prepraed KSCN solution was pipetted into a 100mL flask and the volume was brought up to 100.0 mL with distilled water.
Then, five solutions were prepared according to the chart.
Test Tube #
0.200 M Fe(NO3)3 (mL)
Diluted KSCN (mL)
H2O (mL)
1
5.0
1.0
4.0
2
5.0
2.0
3.0
3
5.0
3.0
2.0
4
5.0
4.0
1.0
5
5.0
5.0
0
6 (blank)
5.0
0.0
5.0
Find the concentration of Fe(SCN)2+ for each test tube.
Find the concentration of Fe(SCN)2+ for each test tube.
30mL of 0.200M Fe(NO3)3 was prepared. Then, 15mL of 0.0020M KSCN was prepraed in another beaker. 10.0mL of prepraed KSCN solution was pipetted into a 100mL flask and the volume was brought up to 100.0 mL with distilled water.
Then, five solutions were prepared according to the chart.
| Test Tube # | 0.200 M Fe(NO3)3 (mL) | Diluted KSCN (mL) | H2O (mL) |
| 1 | 5.0 | 1.0 | 4.0 |
| 2 | 5.0 | 2.0 | 3.0 |
| 3 | 5.0 | 3.0 | 2.0 |
| 4 | 5.0 | 4.0 | 1.0 |
| 5 | 5.0 | 5.0 | 0 |
| 6 (blank) | 5.0 | 0.0 | 5.0 |
Find the concentration of Fe(SCN)2+ for each test tube.
Lelia LubowitzLv2
6 Apr 2019