1
answer
0
watching
533
views
11 Nov 2019
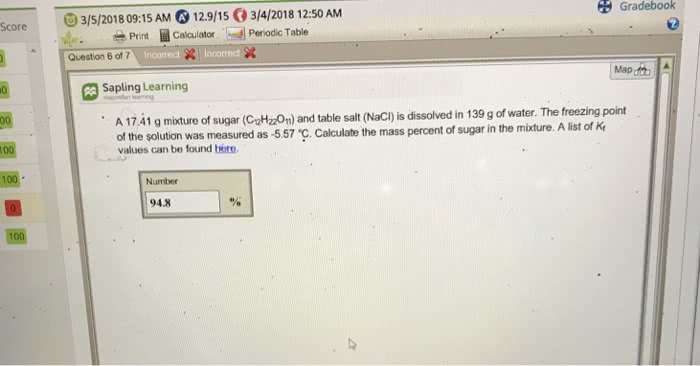
A 14.30 g mixture of sugar (C12H22O11) and table salt (NaCl) is dissolved in 223 g of water. The freezing point of the solution was measured as -2.63 °C. Calculate the mass percent of sugar in the mixture. With the Kf value of water being 1.86C/m
A 14.30 g mixture of sugar (C12H22O11) and table salt (NaCl) is dissolved in 223 g of water. The freezing point of the solution was measured as -2.63 °C. Calculate the mass percent of sugar in the mixture. With the Kf value of water being 1.86C/m
Elin HesselLv2
10 Feb 2019