1
answer
0
watching
274
views
11 Nov 2019

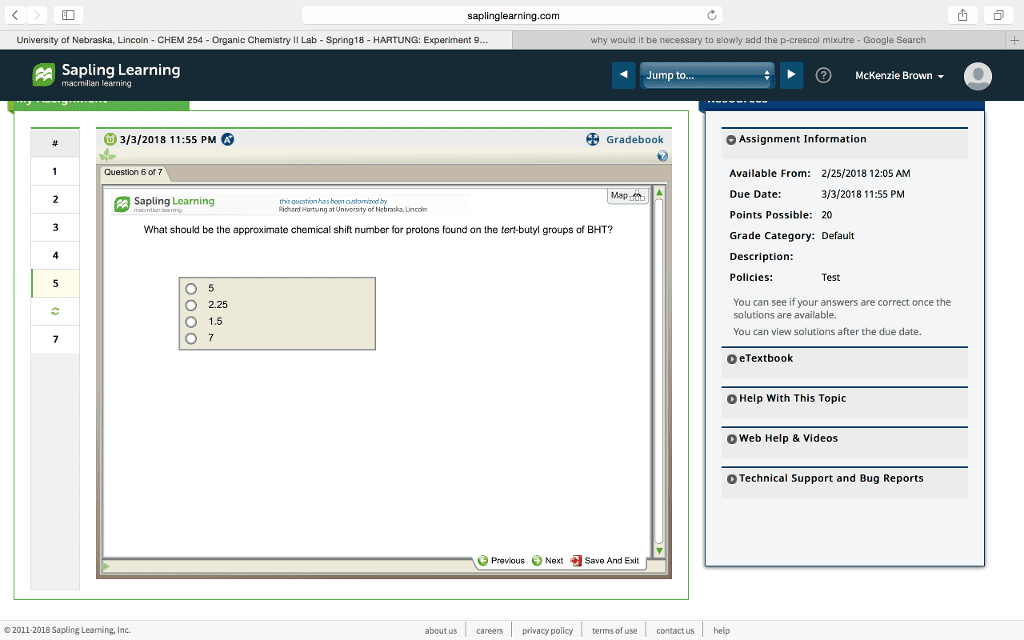
saplinglearning.com University of Nebraska, Lincoln - CHEM 254 - Organic Chemistry II Lab-Spring18- HARTUNG: Experiment 9 why would it be necessary to slowly add the p-crescol mixutre-Google Search Sapling Learning macmillan learning Jump to. ?) McKenzie Brown 3/3/2018 11:55 PM Gradebook Assignment Information Question 5 of 7 le From: 2/25/2018 12:0s Map Due Date: 3/3/2018 11:55 PM Sapling Learning this question has boon customized by idhard Hartung at University of Nebraska, Lincoir Points Possible: 20 A student used 3.945 g of p-cresol and 2.367 g of t-butanol in the synthesis of BHT Which is the limiting reagent and how many moles of BHT can be formed? Grade Category: Default Description: Policies: Test butanol, 0.016 mole of BHT p-cresol, 0.036 mole of BHT p-cresol, 0.018 mole of BHT +butanol, 0.032 mole of BHT You can see if your answers are correct once the solutions are available. You can view solutions after the due date. O O eTextbook Help With This Topic Web Help & Videos Technical Support and Bug Reports Previous Next Save And Exit © 2011-2018 Sapling Learning, Inc. about us careers privacy policy terms of use contact us help

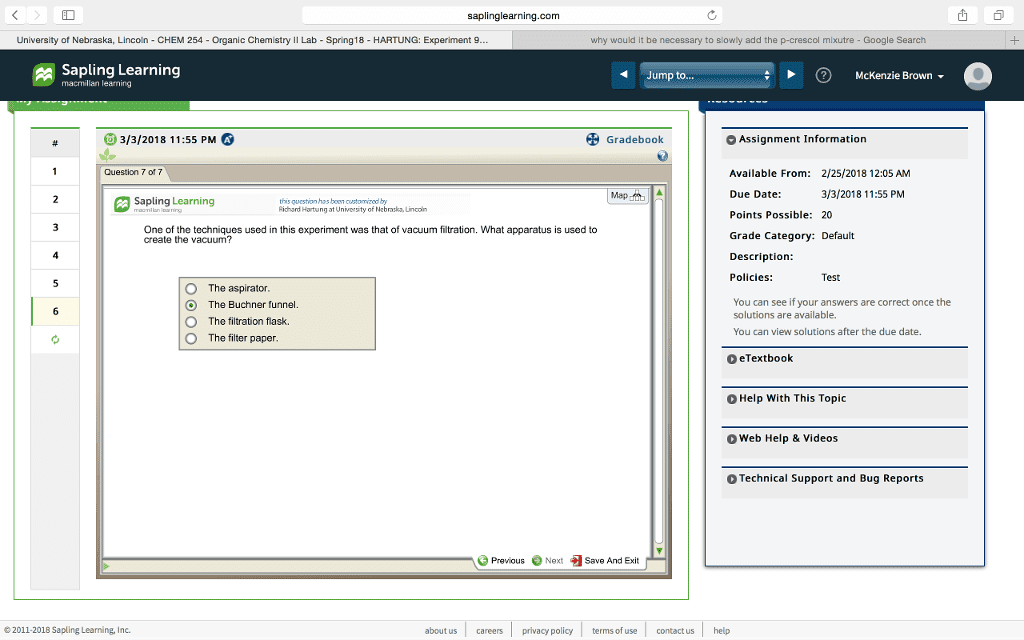
saplinglearning.com University of Nebraska, Lincoln - CHEM 254 - Organic Chemistry II Lab-Spring18- HARTUNG: Experiment 9 why would it be necessary to slowly add the p-crescol mixutre-Google Search Sapling Learning macmillan learning Jump to. ?) McKenzie Brown 3/3/2018 11:55 PM Gradebook Assignment Information Question 5 of 7 le From: 2/25/2018 12:0s Map Due Date: 3/3/2018 11:55 PM Sapling Learning this question has boon customized by idhard Hartung at University of Nebraska, Lincoir Points Possible: 20 A student used 3.945 g of p-cresol and 2.367 g of t-butanol in the synthesis of BHT Which is the limiting reagent and how many moles of BHT can be formed? Grade Category: Default Description: Policies: Test butanol, 0.016 mole of BHT p-cresol, 0.036 mole of BHT p-cresol, 0.018 mole of BHT +butanol, 0.032 mole of BHT You can see if your answers are correct once the solutions are available. You can view solutions after the due date. O O eTextbook Help With This Topic Web Help & Videos Technical Support and Bug Reports Previous Next Save And Exit © 2011-2018 Sapling Learning, Inc. about us careers privacy policy terms of use contact us help
Jamar FerryLv2
5 Jul 2019