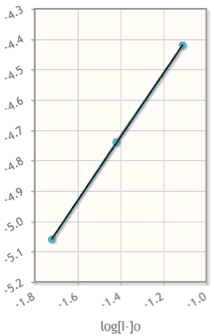
1
answer
0
watching
356
views
11 Nov 2019
1. Calculate and tabulate [I-] in mol/L, [S2O8] in mol/L and initial rate in mol/(L)(s). Account for dilution.
2. Determine the rate law for the reaction of iodide and persulfate ions.
3. Calculate the value of k and its units.
Experiment 1 at 21.2 degrees Celsius
10.0 mL of 0.10 M [KI] (aq) 10.0 mL of 0.10 M [K2S2O8] (aq) 5.0 mL of 0.0050 M [Na2S2O3] (aq) 1.0 mL of starch (aq)
Trial 1 - 97 seconds
Trial 2- 102 seconds
Trial 3 - 95 seconds
1. Calculate and tabulate [I-] in mol/L, [S2O8] in mol/L and initial rate in mol/(L)(s). Account for dilution.
2. Determine the rate law for the reaction of iodide and persulfate ions.
3. Calculate the value of k and its units.
Experiment 1 at 21.2 degrees Celsius
10.0 mL of 0.10 M [KI] (aq)
10.0 mL of 0.10 M [K2S2O8] (aq)
5.0 mL of 0.0050 M [Na2S2O3] (aq)
1.0 mL of starch (aq)
Trial 1 - 97 seconds
Trial 2- 102 seconds
Trial 3 - 95 seconds
1
answer
0
watching
356
views
For unlimited access to Homework Help, a Homework+ subscription is required.
Jarrod RobelLv2
1 Jun 2019
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232