1
answer
0
watching
313
views
11 Nov 2019
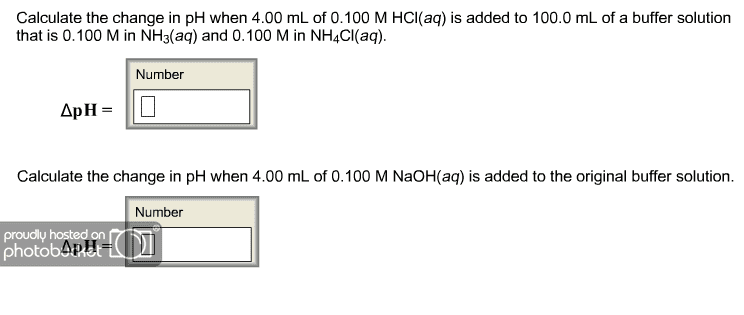
d. You a buffer with 4.00 M NH3 and 0.600 M NH4Cl
i. What is the pH of this buffer solution? Kb = 1.8 Ã 10-5 (2 Marks)
ii. To 100.0 mL of this buffer solution is added 25.0 mL of 0.300 M HCl, what is the pH of the resulting solution (4 Marks)
iii. To 200.0 mL of this buffer solution is added 40.0 mL of 0.300 M NaOH, what is the pH of the resulting solution (4 Marks)
2)2.50 g of HClO was dissolved in 250 mL of water
a) Calculate the pH
b) Calculate pOH
d. You a buffer with 4.00 M NH3 and 0.600 M NH4Cl
i. What is the pH of this buffer solution? Kb = 1.8 Ã 10-5 (2 Marks)
ii. To 100.0 mL of this buffer solution is added 25.0 mL of 0.300 M HCl, what is the pH of the resulting solution (4 Marks)
iii. To 200.0 mL of this buffer solution is added 40.0 mL of 0.300 M NaOH, what is the pH of the resulting solution (4 Marks)
2)2.50 g of HClO was dissolved in 250 mL of water
a) Calculate the pH
b) Calculate pOH
Lelia LubowitzLv2
4 Nov 2019