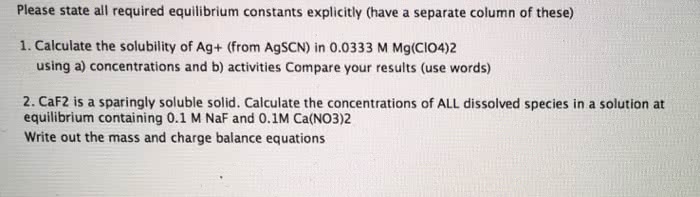
1
answer
0
watching
268
views
11 Nov 2019
A Precipitate of CaF2 has been formed in a solution containing 0.025 M NaNO3 and 0.015 M Ca(NO3)2.
(a) Calculate the inonic strength of the solution. (At this point you may neglect the contribution from the ions of the precipitate.)
(b) Using the activity coefficients, calculate the molar solubility of CaF2 in this solution. Thermodynamic Ksp for CaF2=3.9x10-11 (HINT: you will have to carry out an interpolation.)
(c) Verify the assumption in (a): [Ca2+] and [F-] from CaF2 are negligible. If you made any assumptions in (b), verify them as well.
(d) Calculate the mass, in grams, of F- (i.e. the mass of the ion that will be dissolved) that will be in 250.0 mL of this solution at equilibrium.
A Precipitate of CaF2 has been formed in a solution containing 0.025 M NaNO3 and 0.015 M Ca(NO3)2.
(a) Calculate the inonic strength of the solution. (At this point you may neglect the contribution from the ions of the precipitate.)
(b) Using the activity coefficients, calculate the molar solubility of CaF2 in this solution. Thermodynamic Ksp for CaF2=3.9x10-11 (HINT: you will have to carry out an interpolation.)
(c) Verify the assumption in (a): [Ca2+] and [F-] from CaF2 are negligible. If you made any assumptions in (b), verify them as well.
(d) Calculate the mass, in grams, of F- (i.e. the mass of the ion that will be dissolved) that will be in 250.0 mL of this solution at equilibrium.
Tod ThielLv2
16 Apr 2019