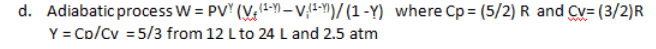1
answer
0
watching
72
views
11 Nov 2019
Can you help me solve this:
Please show the work, and steps. I really dont know how to do it. Thank you
.-Calculate the Heat and the work transferred in a process at:
a)Constant temperature of 350 K when 2.5 moles of The volume of a monoatomic ideal gas increased from 10 L to 30 L. QT = WT = nRT ln ( Vf/Vi)
b)At constant pressure of 1.5 atm and 343 K when the volume of an ideal gas increased from 15 L to 55 L
c)At constant pressure Wp = P ( Vfinal â Vinitial)
d)Adiabatic process W = PVá»´ (Vf(1-á»´) â Vi(1-á»´))/ (1 -á»´) where Cp = (5/2) R and Cv= (3/2)R
á»´ = Cp/Cv = 5/3 from 12 L to 24 L and 2.5 atm
Can you help me solve this:
Please show the work, and steps. I really dont know how to do it. Thank you
.-Calculate the Heat and the work transferred in a process at:
a)Constant temperature of 350 K when 2.5 moles of The volume of a monoatomic ideal gas increased from 10 L to 30 L. QT = WT = nRT ln ( Vf/Vi)
b)At constant pressure of 1.5 atm and 343 K when the volume of an ideal gas increased from 15 L to 55 L
c)At constant pressure Wp = P ( Vfinal â Vinitial)
d)Adiabatic process W = PVá»´ (Vf(1-á»´) â Vi(1-á»´))/ (1 -á»´) where Cp = (5/2) R and Cv= (3/2)R
á»´ = Cp/Cv = 5/3 from 12 L to 24 L and 2.5 atm
Nelly StrackeLv2
18 Jul 2019