1
answer
0
watching
115
views
11 Nov 2019
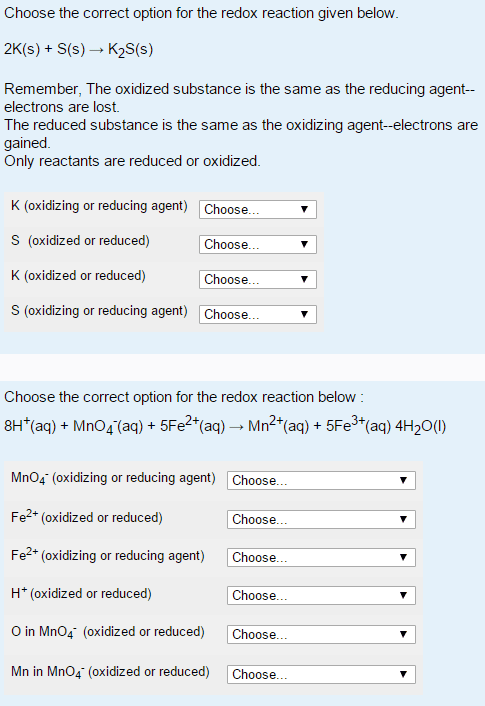
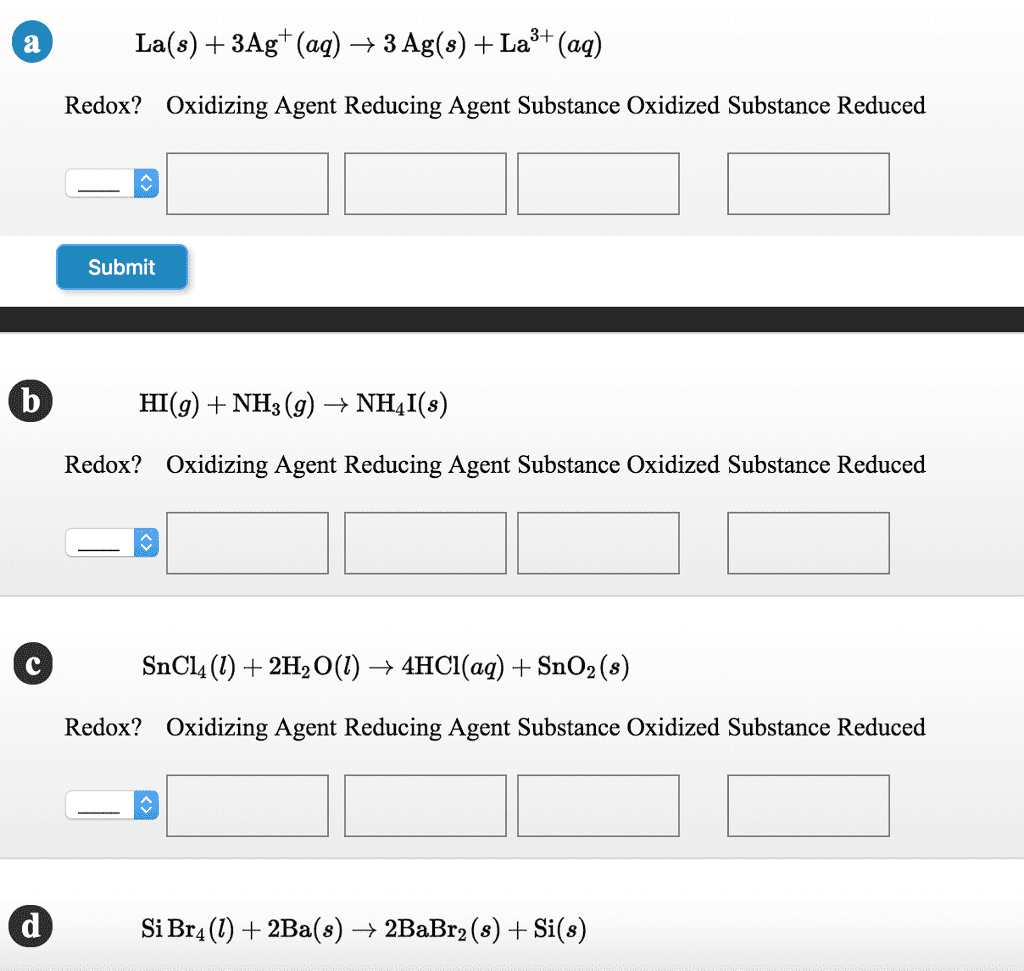
Answer the following questions concerning the reaction below:
HSO3Â- (aq) + I2(s) + H2O (l) â HSO4Â-(aq) + 2 HI (aq)
a) What element is being reduced in this reaction?
I , O, H, s
b) What element is bieng oxidized in this reaction?
S, I,O,H
c) What is the oxidizing agent in this reaction?
HI, H, S, O, HSO3-
HSO4-, H20, I, I2
d)What is the reducing agent in this reaction?
HSO3-, H2O, HI, S
H, O, I, I1, HSO4-
Answer the following questions concerning the reaction below:
HSO3Â- (aq) + I2(s) + H2O (l) â HSO4Â-(aq) + 2 HI (aq)
a) What element is being reduced in this reaction?
I , O, H, s
b) What element is bieng oxidized in this reaction?
S, I,O,H
c) What is the oxidizing agent in this reaction?
HI, H, S, O, HSO3-
HSO4-, H20, I, I2
d)What is the reducing agent in this reaction?
HSO3-, H2O, HI, S
H, O, I, I1, HSO4-
Keith LeannonLv2
17 May 2019