1
answer
0
watching
568
views
12 Nov 2019
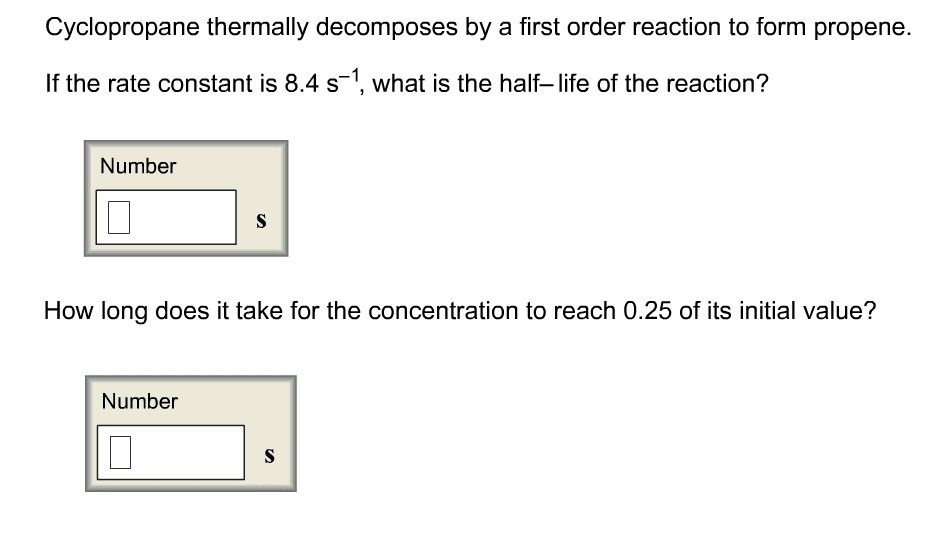
a) Cyclopropane thermally decomposes by a first order reaction to form propene. If the rate constant is 8.8 sâ1, what is the halfâlife of the reaction?
b) How long does it take for the concentration to reach 0.50 of its initial value?
a) Cyclopropane thermally decomposes by a first order reaction to form propene. If the rate constant is 8.8 sâ1, what is the halfâlife of the reaction?
b) How long does it take for the concentration to reach 0.50 of its initial value?
Elin HesselLv2
21 Jan 2019