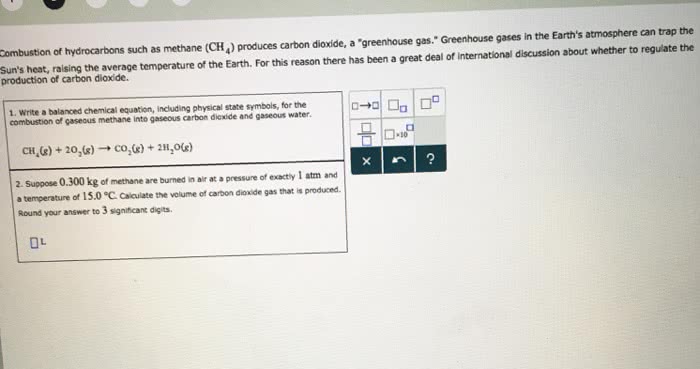
1
answer
0
watching
218
views
12 Nov 2019
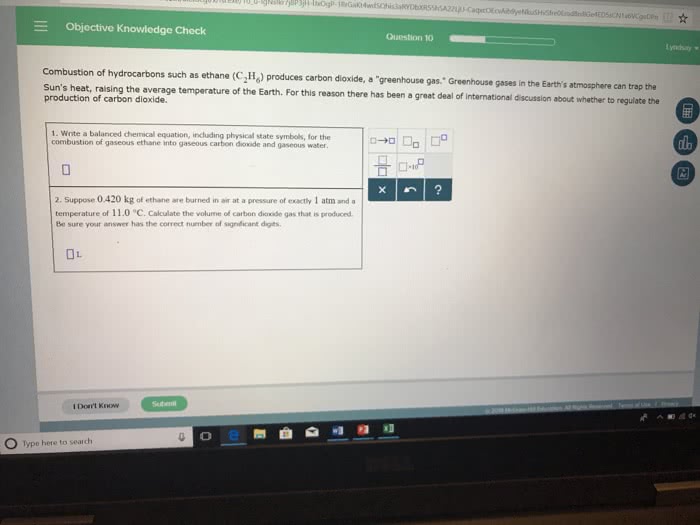

Objective Knowledge Check Question 10 lyndsaty h as ethane (CãHg) produces carbon dioxide, a-greenhouse gas." Greenhouse gases in the Earth's atmosphere can trap the Sun's heat, raising the average temperature of the Earth. For this reason there has been a great deal of production of carbon dioxide. international discussion about whether to regulate the 1. Write a balanced chemical equation, including physical state symbols, for the combustion of gaseous ethane into gaseous carbon dioxide and gaseous water 2. Suppose 0.420 kg of ethane are burned in air at a pressure of exactly 1 atm and a temperature of 11.0 °C. Calculate the volume of carbon dioxide gas that is produced Be sure your answer has the correct number of signiéicant digits I Don't Know o e O Type here to search
Jarrod RobelLv2
12 Sep 2019