1
answer
0
watching
1,041
views
12 Nov 2019
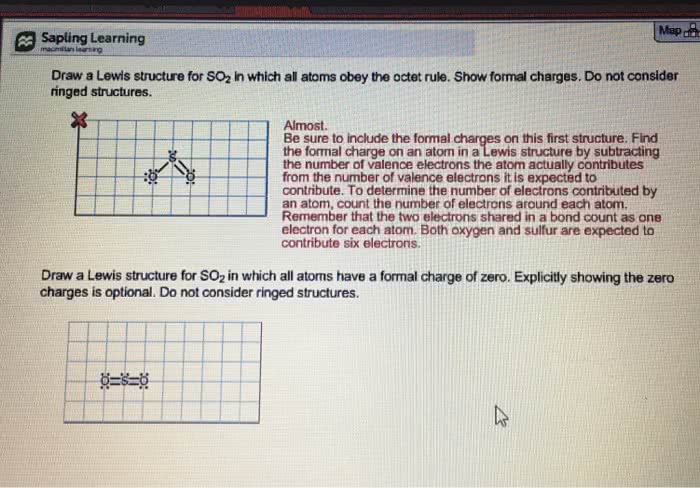
Question 1 . http://www.tiikoni.com/tis/view/?id=94b4e52
Question 2. white phosphorous has the chemical formula P4(s). A P4 molecule has 20 valence electrons. Draw a Lewis formula for a white phosphorous molecule in which none of the atoms violates the octet rule and the formal charge on each atom is zero. There are no pi bonds in the structure.
Question 1 . http://www.tiikoni.com/tis/view/?id=94b4e52
Question 2. white phosphorous has the chemical formula P4(s). A P4 molecule has 20 valence electrons. Draw a Lewis formula for a white phosphorous molecule in which none of the atoms violates the octet rule and the formal charge on each atom is zero. There are no pi bonds in the structure.
Elin HesselLv2
20 Jan 2019