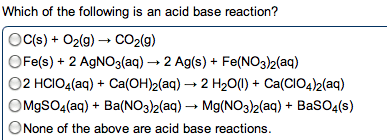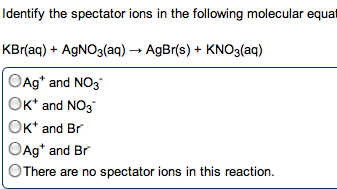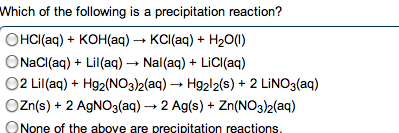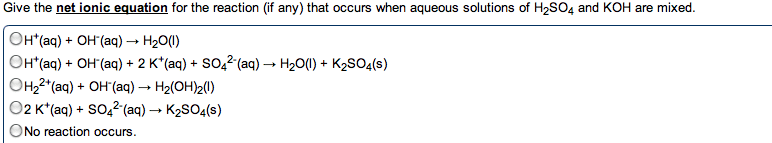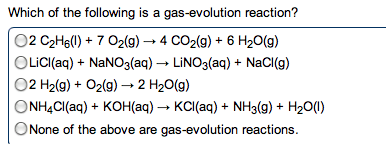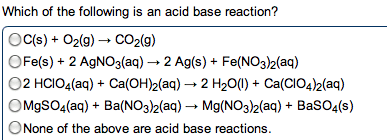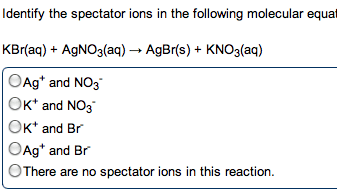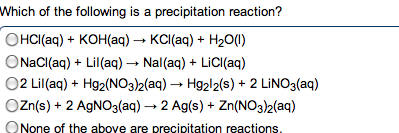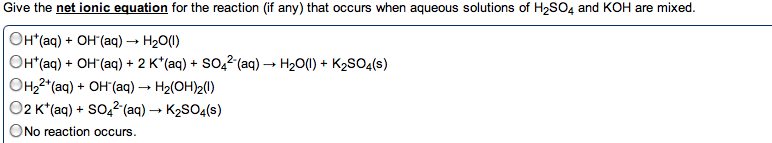2
answers
1
watching
4,135
views
28 Sep 2019
give the net ionic equation for the reaction (if any) that occurs when aqueous solutions K2S and Fe(NO3)2 are mixed...Sooooo......K2S + Fe(NO3)2----> ?????? K2(NO3)2 +FeS2?? I really need help figuring out if this last part is right I don't understand is the subscript 2 after no3 the charge? the number of atoms? please help
give the net ionic equation for the reaction (if any) that occurs when aqueous solutions K2S and Fe(NO3)2 are mixed...Sooooo......K2S + Fe(NO3)2----> ?????? K2(NO3)2 +FeS2?? I really need help figuring out if this last part is right I don't understand is the subscript 2 after no3 the charge? the number of atoms? please help
2
answers
1
watching
4,135
views
For unlimited access to Homework Help, a Homework+ subscription is required.
desmarcos19Lv10
17 Jan 2022
Beverley SmithLv2
28 Sep 2019
Already have an account? Log in
Related textbook solutions
Basic Chemistry
5 Edition,
Timberlake
ISBN: 9780134138046
Principles of Chemistry Molecular Approach
4th Edition,
Tro
ISBN: 9780134112831
Principles of Chemistry Molecular Approach
3rd Edition, 2014
Tro
ISBN: 9780321971944
Chemistry: Structure and Properties
2nd Edition,
Tro
ISBN: 9780134293936
Chemistry: A Molecular Approach
3rd Edition,
Tro
ISBN: 9780321809247
Chemistry: A Molecular Approach
5th Edition,
Tro
ISBN: 9780134874371
Principles of Chemistry: A Molecular Approach
4th Edition,
Tro
ISBN: 9780134895741
Chemistry: The Central Science
14th Edition, 2017
Brown
ISBN: 9780134414232